Fill in any nonbonding valence electrons that are missing from the followingstructures: CH (b) (c) (a)
Question:
Fill in any nonbonding valence electrons that are missing from the followingstructures:
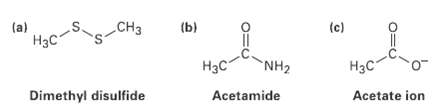
Transcribed Image Text:
CHз (b) (c) (a) Нас" NH2 Нас- Нас Dimethyl disulfide Acetate ion Acetamide
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 42% (14 reviews)
a 00 H...View the full answer

Answered By

JAPHETH KOGEI
Hi there. I'm here to assist you to score the highest marks on your assignments and homework. My areas of specialisation are:
Auditing, Financial Accounting, Macroeconomics, Monetary-economics, Business-administration, Advanced-accounting, Corporate Finance, Professional-accounting-ethics, Corporate governance, Financial-risk-analysis, Financial-budgeting, Corporate-social-responsibility, Statistics, Business management, logic, Critical thinking,
So, I look forward to helping you solve your academic problem.
I enjoy teaching and tutoring university and high school students. During my free time, I also read books on motivation, leadership, comedy, emotional intelligence, critical thinking, nature, human nature, innovation, persuasion, performance, negotiations, goals, power, time management, wealth, debates, sales, and finance. Additionally, I am a panellist on an FM radio program on Sunday mornings where we discuss current affairs.
I travel three times a year either to the USA, Europe and around Africa.
As a university student in the USA, I enjoyed interacting with people from different cultures and ethnic groups. Together with friends, we travelled widely in the USA and in Europe (UK, France, Denmark, Germany, Turkey, etc).
So, I look forward to tutoring you. I believe that it will be exciting to meet them.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Fill in any unshared electron pairs that are missing from the following formulas: (a) (CH3CH2)2NH (b) (c) CH3CH2SCH2CH3 (d) CH3OCH2CH2OH CH, O=U
-
Fill in the reagents a that are missing from the followingscheme: CC -CH CH . H ,
-
Certain item descriptions and amounts are missing from the monthly schedule of cost of goods manufactured below and the income statement of Tretinik Manufacturing. Fill in the missing items. TRETINIK...
-
What are the advantages and disadvantages of seismic reflection data sets?
-
Blue Company, an architectural firm, has a bookkeeper who maintains a cash receipts and disbursements journal. At the end of the year (2016), the company hires you to convert the cash receipts and...
-
The Fries rearrangement occurs when a phenolic ester is heated with a Friedel-Crafts catalyst such as AlCl3: The reaction may produce both ortho and para acylated phenols, the former generally...
-
A simplified schematic diagram of a carburetor of a gasoline engine is shown in Fig. P11.38. The throat area is \(0.5 \mathrm{in}^{2}\). The engine draws air downward through the carburetor Venturi...
-
Adams, Peters, and Blake share profits and losses for their APB Partnership in a ratio of 2:3:5. When they decide to liquidate, the balance sheet is as follows: Liquidation expenses are expected to...
-
Find an acquaintance willing to watch a short clip of a car crash (it is no worse than what you'd see on Mythbusters): Tell them this explicitly - "My professor is asking us to do the assignment to...
-
Bonnie and Clyde want to take a six-month leave of absence from their jobs to travel extensively in South America. Rounded to the next higher month, how long will it take them to save $40,000 for the...
-
Draw a line-bond structure for vinyl chloride, C2H3C1, the starting material from which PVC [poly (vinyl chloride)] plastic is made.
-
Convert the following line-bond structures into molecularformulas: CH CH- (a) (b) . Hi H. c=C Vitamin C Aspirin (acetylsalicylic acid) (ascorbic acid) (c) H. - (d) H~ CH c-o H. . HOI I H H OH...
-
Comparative financial statements for Pen Corporation and its subsidiaries, Sir and Tip Corporations, for the year ended December 31, 2011, are as follows (in thousands): ADDITIONAL INFORMATION1. Pen...
-
Kay owns two annuities that will each pay $500 a month for the next 12 years. One payment is received at the beginning of each month while the other is received at the end of each month. At a...
-
Why is it difficult to addresses a variety of readers who have different perspectives from those of the writer? What specific actions should you take to lessen or eliminate this issue?
-
Macys made 2 distributions to shareholders this year: Mar. 15: $120,000 and Sep 15 $80,000. Macys had current E&P of $50,000 and accumulated E&P of $100,000. How will this E&P be allocated to the...
-
Consider the following high-level code snippet. Assume that the base addresses of arrayl and array2 are held in R1 and R2 and that array2 is initialized before it is used. int i; int array1[100]; int...
-
What is the organization and citation style for journal articles in criminal justice? Why do you think the organization of papers in criminal justice makes sense for that field? What academic or...
-
The most appropriate risk attribution approach for the fixed-income manager is to: A. decompose historical returns into a top-down factor framework. B. evaluate the marginal contribution to total...
-
Choose two matrices A and B with dimension 2 x 2. Calculate det A, det B, and det (AB). Repeat this process until you are able to discover how these three determinants are related. Summarize your...
-
Calculate the vapor pressure of n-decane as a function of temperature using the Peng-Robinson equation of state. Compare your results with (a) Literature values (b) Predictions using the...
-
Bond strengths can be used to estimate the relative stability of isomers that have different bonds. The isomer that has the larger total bond energy is more stable. One of the following isomers is...
-
Bond strengths can be used to estimate the relative stability of isomers that have different bonds. The isomer that has the larger total bond energy is more stable. One of the following isomers is...
-
One of the isomers of C 5 H 12 reacts with Cl 2 in the presence of light to produce three isomers of C 5 H 11 Cl: This reaction replaces am one of the hydrogen?s of C 5 H 12 with a Cl. What arc the...
-
Whatare basis of the segmentation? How would you improve on this strategy in marketing ? What are the basis of the segmentation? How would you improve on this strategy?
-
Jimmy has a bond with a $1,000 face value and a coupon rate of 9.5% paid semiannually. It has a five-year life. a) If investors are willing to accept a 14 percent rate of return on bonds of similar...
-
A zero-coupon bond that will pay $1,050 in 12 years is selling today for $441.91. What interest rate does the bond offer?

Study smarter with the SolutionInn App


