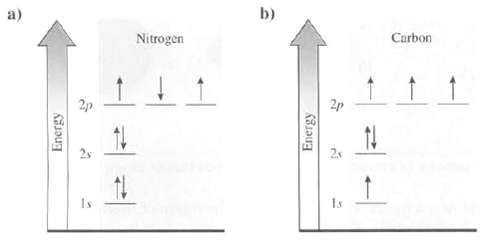
Explain whether the electron arrangement for these atoms is the ground state or an excitedstate: a) Energy
Question:
Explain whether the electron arrangement for these atoms is the ground state or an excitedstate:
Transcribed Image Text:
a) Energy 3 Nitrogen b) Energy 2p 5 4 ↑ Carbon
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (11 reviews)
The electron arrangement that is in accord with the basic rul...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
An electron is in the ground state in a two-dimensional, square, infinite potential well with edge lengths L. We will probe for it in a square of area 400 pm2 that is centered at x = L/8 and y = L/8....
-
For an electron in the ground state of a hydrogen atom, calculate its (a) Potential energy, (b) Kinetic energy, and (c) Total energy.
-
An electron is in the ground state in a two-dimensional, square, infinite potential well with edge lengths L. We will probe for it in a square of area 400 pm2 that is centered at x = L/8 and y = L/8....
-
It is stated in Section 40.3 that a finite potential well always has at least one bound level, no matter how shallow the well. Does this mean that as U 0 0, E 1 0? Does this violate the Heisenberg...
-
Rabbid Industries Ltd consists of three decentralized divisions: Brentwood Division, Crater Division and Dollar Division. The managing director of Rabbid Industries has given the managers of the...
-
The file MutualFunds contains a data set with information for 45 mutual funds that are part of the Morningstar Funds 500. The data set includes the following five variables: Fund type: The type of...
-
Stephen recognizes that there are different techniques for attracting attention to advertisements, and he wants to test three of these for the sample product: all color, all black-and-white, and a...
-
Pal Corporation acquired 70 percent of the outstanding voting stock of Sal Corporation for $91,000 cash on January 1, 2011, when Sal's stockholders' equity was $130,000. All the assets and...
-
Your company is monitoring Azure virtual machines by using the Microsoft Defender for Cloud. What should you use to configure which applications can run on virtual machines?
-
A ride hailing company has their DB structured in 3 major tables as described in the SCHEMA section below. Write a query to fetch the top 100 users who traveled the most distance using the service....
-
Show an atomic orbital energy level diagram for these atoms: (a) Si (b) Al (c) Cl
-
Show an energy level diagram for the MOs for He 2 and show how the electrons would be arranged in these MOs.
-
Use the data given in the wind rose Figure 28.19 To make a crude estimate of the average ?v? and root mean cube v? wind speed at Klamath Lake, Oregon. From these, estimate the Weibull parameters for...
-
Answer each question in a thorough, well-supported and explained response. 1. Define demand. What does the law of demand state? Express it in words, in symbols (clip art or images), and as a graph....
-
1. 2. 3. Suppose that the long-run world demand and supply elasticities of crude oil are -0.906 and 0.515, respectively. The current long-run equilibrium price is $30 per barrel and the equilibrium...
-
United Overseas Airline (UOA)Company The United Overseas Airline Co. was founded five years ago by three Airline employees of TWA and Pan American airlines Company employees. The three founders own...
-
1. Entrepreneurial Perks: A risk-averse entrepreneur is considering selling stock in her company to the public. She will continue to manage the firm after it "goes public". The entrepreneur gets...
-
Which number is A equal to? A/ (Please enter a numercial value. For example, to enter "three thousand" you need to type in "3000")
-
If Exercise 8.3 had asked about a book in free fall rather than one on the floor, what would the free-body diagram look like? Data from Exercise 8.3 Draw a free-body diagram for a book lying...
-
The Thomas Corporation was organized on Jan. 1, 2020. On Dec. 31, 2021, the corporation lost most of its inventory in a warehouse fire before the year-end count of inventory was to take place. just...
-
What is wrong with the following calculation? 3 (-4) --- 1 --4 dx = = -1 X 1 3 3
-
Starting from testosterone (Problem 17.48), how would you prepare the followingsubstances? CH (a) (b) C CH - (c) (d) CH CH -
-
Compound A, C10H18O, undergoes reaction with dilute H2SO4 at 25 C to yield a mixture of two alkenes, C10H16. The major alkene product, B, gives only Cyclopentanone after ozone treatment followed by...
-
Dehydration of trans-2-methylcyclopentanol with POCl3 in pyridine yields predominantly 3-methylcyclopentene. Is the stereochemistry of this dehydration syn or anti? Can you suggest a reason for...
-
Compute the estimated inventory at May 31, assuming that the gross profit is 30% of sales. 5. (5 points) The records of Air Bud's Boutique report the following data for the month of April. Purchases...
-
1. What is a forward contract? 2. Why do you need fx swap ? 3. If the rate is usd to cad, then you multiple by the rate. If converting USD to CAD. If the rate is CAD to USD, then you have to divide...
-
The number 81 64. 1/4 is equivalent to:

Study smarter with the SolutionInn App


