Explain which mechanism is preferred in these reactions and show the majorproducts: Br b) 1-BUOH 0- PROH
Question:
Explain which mechanism is preferred in these reactions and show the majorproducts:
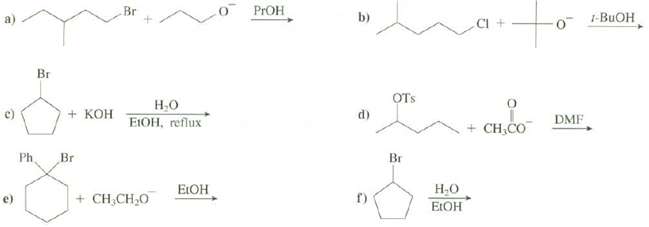
Transcribed Image Text:
Br b) 1-BUOH 0- PROH Br Н.о EIOH, reflux OTs d) DMF + CH,CO + KOH Ph Br Br Н.о ELOH ELOH f) e) + CH,CH,0
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 64% (17 reviews)
a The leaving group is on a primary carbon and the base is not sterically hindere...View the full answer

Answered By

Douglas Makokha
Unlock Academic Success with Dedicated Tutoring and Expert Writing Support!
Are you ready to excel in your academics? Look no further! As a passionate tutor, I believe that dedication and hard work are the keys to achieving outstanding results. When it comes to academics, I strive to provide nothing but the best for every student I encounter.
With a relentless thirst for knowledge, I have extensively researched numerous subjects and topics, equipping myself with a treasure trove of answers to tackle any question that comes my way. With four years of invaluable experience, I have mastered the art of unraveling even the most intricate problems. Collaborating with esteemed writers has granted me exclusive access to the trade secrets utilized by the industry's top professionals.
Allow me the pleasure of assisting you with your writing assignments. I thrive on challenges and will guide you through any obstacles you may face. Together, we will unlock your academic potential and pave the way for your success.
4.90+
60+ Reviews
341+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Show how you would use Suzuki reactions to synthesize these products from the indicated starting materials. You may use any additional reagents you need. (a) (b) Br
-
In contrast to SN2 reactions, SN1 reactions show relatively little nucleophile selectivity. That is, when more than one nucleophile is present in the reaction medium, SN1 reactions show only a slight...
-
Show that bimolecular reactions between nonlinear molecules are much slower than between atoms even when the activation energies of both reactions are equal. Use transition state theory and make the...
-
Building a Case Study Analysis on Outsourcing, Find a minimum of three news articles discussing this issue, prepare the case study report that has three main parts: Summary and Overview of the issue...
-
A discussion of product costing systems is irrelevant to our firm as we only produce services, not products. Discuss.
-
What are the four objectives in the strategic role of cost allocation? Explain each briefly.
-
Show that the stochastic process \[e^{\int_{0}^{t} c(s) d B_{s}-\frac{1}{2} \int_{0}^{t} c^{2}(s) d s}\] is a martingale for any deterministic function \(c(t)\). Does the result change if \(c(t,...
-
1. What is the difference between rules-based and principles-based accounting standards, and what are the advantages and disadvantages of each? 2. Why has U.S. GAAP evolved into a rules-based...
-
Mountain Air Limited manufactures a line of room air purifiers. Management is currently evaluating the possible production of an air purifier for automobiles. Based on an annual volume of 1 0 , 0 0 0...
-
1. In what respects is the change consistent with Alfred Chandlers phrase, structure follows strategy? 2. Why do investors think that the new Alphabet structure is worth an extra $29 billion?
-
Show the substitution and/or elimination products for these reactions. Explain which mechanism are occurring and which product you expect to be the majorone. OTs a) CH,CH,CHCH, + CH,CH,CO CH,CH,CO,H...
-
Explain which reaction mechanism (E1, SN1, E2, SN2) these reactions follow, and show the majorproducts: Br a) DMSO + CN Br b) . reflux + OH Br CH c) EIO LOTS + NaOEt d) t-BUOH CI + t-BUOK e) . Br CH,...
-
Working backward to derive proceeds from disposition of plant assets. The balance sheets of Wilcox Corporation at the beginning and end of the year contained the following data: During the year,...
-
What is compounding of an impulse turbine? State the principle of working of an open-cycle gas turbine. What are the advantages of gas turbines over Steam turbines?
-
Explain how the net interest margin (NIM) would respond to increased competition for funds by the financial intermediation industry.
-
Write the function of the following: (a) Nozzle (b) Moving blade (c) Guide blades in steam turbine
-
Explain the working of closed cycle gas turbine.
-
In a gas turbine cycle, the condition of air at the entrance of compressor is 1 bar and \(27^{\circ} \mathrm{C}\). Pressure ratio is 6 . Maximum temperature is \(700^{\circ} \mathrm{C}\). The exhaust...
-
Generate the Janecke diagram and the distribution curve for the system cottonseed oil (A)-propane (B)-oleic acid (C) at \(372 \mathrm{~K}\) and \(42.5 \mathrm{~atm}\).
-
How can NAFTA be beneficial to suppliers of Walmart?
-
Find the mode, median, mean, lower quartile, upper quartile, and interquartile range for eacl data set. 6.5 8 5.5 10 9.5 7 Shoe Size 7.5 6 5.5 10 6 7.5 9 6 9 9
-
How many peaks would you expect in the 1H NMR spectrum of 1, 4-dimethyl-benzene para-xy1ene or p-xylene)? What ratio of peak areas would you expect on integration of the spectrum? Refer to Table 13.3...
-
Predict the splitting patterns you would expect for each proton in the followingmolecules: (b) CH2H2Br (a) CHBr2CH3 (c) CICH,CH2CH2CI (e) CHCH-CHCH (f) (d) CHCH2CH CH ondeo CH
-
Draw structures for compounds that meet the following descriptions: (a) C2H6O; one singlet (b) C3H7Cl; one doublet and one septet (c) C4H8Cl2O; two triplets (d) C4H8O2, one singlet, one triplet, and...
-
A couple obtained a $20,000 mortgage loan at an interest rate of 10.5% compounded monthly. (Original principal equals to PV of all payments discounted at the interest rate on the loan contract) (1)...
-
What strategies and tactics are employed to manage strategic risks and uncertainties, including geopolitical instability, supply chain disruptions, and emerging competitive threats, while preserving...
-
How do strategic planners integrate ethical considerations and sustainability imperatives into strategic planning processes, balancing short-term financial objectives with long-term societal and...

Study smarter with the SolutionInn App


