How many peaks would you expect in the 1H NMR spectrum of 1, 4-dimethyl-benzene para-xy1ene or p-xylene)?
Question:
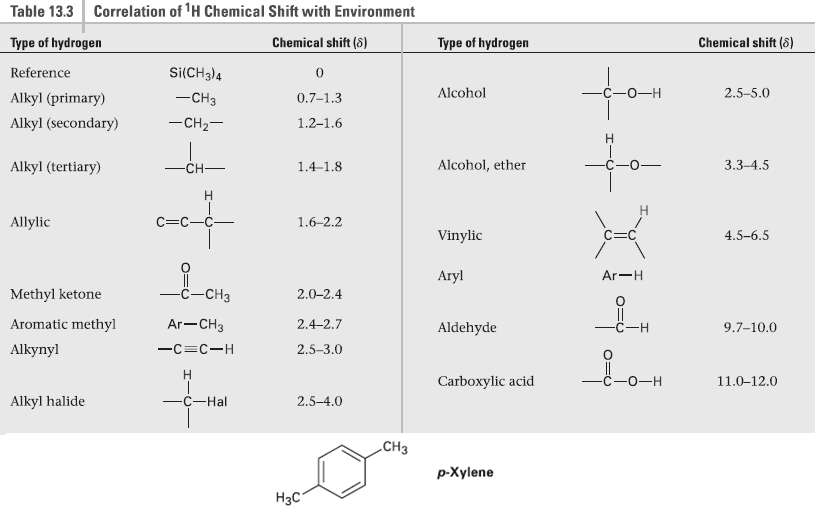
How many peaks would you expect in the 1H NMR spectrum of 1, 4-dimethyl-benzene para-xy1ene or p-xylene)? What ratio of peak areas would you expect on integration of the spectrum? Refer to Table 13.3 for approximate chemical shifts, and sketch what the spectrum would look like. (Remember from Section 2.4 that aromatic rings have two resonanceforms.)
Transcribed Image Text:
Correlation of 'H Chemical Shift with Environment Table 13.3 Type of hydrogen Chemical shift (8) Chemical shift (8) Type of hydrogen Si(CH3)4 Reference —С -о—н Alcohol 2.5-5.0 -CH3 Alkyl (primary) 0.7-1,3 -CH2- Alkyl (secondary) 1.2-1.6 for H. 3.3-4.5 -CH- Alcohol, ether Alkyl (tertiary) 1.4-1.8 C=C-C- Allylic 1.6-2.2 Vinylic 4.5-6.5 Aryl Ar-H -c-CH3 Methyl ketone -CHз 2.0-2.4 Ar-CH3 Aromatic methyl 2.4-2.7 9.7-10.0 Aldehyde C-H 2.5-3.0 Alkynyl -C=C-H С -о—н Carboxylic acid 11.0-12.0 -C-Hal Alkyl halide 2.5-4.0 СНз p-Xylene Нас
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (20 reviews)
HC CH3 pXylene There are two absorptions in the H ...View the full answer

Answered By

ARCHANA BISHT
I had Completed my M.Sc. from Banaras Hindu University. After that I cleared CSIR NET exam two times and GATE (MA) exam. I started online tutoring on various websites and I had an experience of 4 years. I also worked in an academy for 1 years where I help the students in the preparation of CSIR NET and GATE.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
How many signals would you expect in the 1 H NMR spectrum of each of the following compounds: (a) (b) (c) (d) (e) (f) CI Br -
-
How many peaks would you expect to see on the strip chart after amino acid analysis of bradykinin? Arg-Pro-Pro-Gly-Phe-Ser-Pro-Phe-Arg Bradykinin
-
The 1H NMR spectrum of 1-chloropentane is shown at 60 MHz (spectrum H) and 500 MHz (spectrum I), Explain the differences in appearance of the two spectra, and assign the signals to specific hydrogens...
-
The profit before tax as reflected in the draft statement of comprehensive income of Sword Limited for the financial years ended 31 December 2020 and 31 December 2021 respectively was as follows:...
-
How do you think the equilibrium in question 15 will change if cross-price elasticity's of demand increase? How would you alter the equation to show such an increase? Can you compute the new...
-
Find the solution of the differential equation that satisfies the given boundary condition(s). y'' - 2ky' + k 2 y = 0, k 0, y (0) = 1, y (1) = 0
-
Recall that the density of a gamma distribution with shape parameter \(\alpha\) and scale parameter \(\theta\) has a density given by \(\mathrm{f}(y)=\) \(\left[\theta^{\alpha} \Gamma(\alpha)...
-
Do Brands Have Finite Lives? Often, after a brand begins to slip in the marketplace or disappears altogether, commentators observe, All brands have their day, implying brands have a finite life and...
-
Compute the standard deviation for your two asset portfolio Asset X: Weight = 0.5 Standard deviation = 0.0595 Correlation = 0.3568 Asset Y: Weight = 0.5 Standard deviation = 0.0930 Correlation =...
-
Near the end of 2013, the management of Dimsdale Sports Co., a merchandising company, prepared the following estimated balance sheet for December 31, 2013. To prepare a master budget for January,...
-
Identify the different kinds of non-equivalent protons in the following molecule, and tell where you would expect each toabsorb. CH-CH C
-
Predict the splitting patterns you would expect for each proton in the followingmolecules: (b) CH2H2Br (a) CHBr2CH3 (c) CICH,CH2CH2CI (e) CHCH-CHCH (f) (d) CHCH2CH CH ondeo CH
-
Larry Kwang insists that the costs of his schools fundraising project should be determined after the project is complete. He argues that only after the project is complete can its costs be determined...
-
How does the concept of phenotype encompass the observable traits, characteristics, and behaviors of an organism, integrating both its genotype and environmental influences, and how do phenotypic...
-
What business and ethical reasons are there for encouraging diversity and inclusion? What works when it comes to changing employee attitudes and behaviors toward diversity and inclusion?
-
How would you describe Workforce Diversity? Why is it important for managers to have an understanding of diversity?
-
Describe at least 3 fungal pathogens of humans.
-
The profit function on a product is given by P(x) = -0.01x+4x-142, where a is the number of units sold and produced, and P(z) is the total profit, in thousands of dollars, from selling and producing...
-
You may have heard that most car accidents happen within 25 miles of home. The data provided in Table 2.10 is the mileage away from home for a random sample of 36 car accidents that occurred over the...
-
The diameter of a sphere is 18 in. Find the largest volume of regular pyramid of altitude 15 in. that can be cut from the sphere if the pyramid is (a) square, (b) pentagonal, (c) hexagonal, and (d)...
-
An aqueous solution containing 17.5 g of an unknown molecular (nonelectrolyte) compound in 100.0 g of water has a freezing point of -1.8 C. Calculate the molar mass of the unknown compound.
-
Convert the following infrared wavelengths to (a) 6.24 (m, typical for an aromatic (b) 3.38 (m, typical for a saturated bond (c) 5.58 (m typical for a ketone carbonyl (d) 5.75 (m typical for an ester...
-
All of the following compounds absorb infrared radiation between 1600 and In each case, 1. Show which bonds absorb in this region. 2. Predict the approximate absorption frequencies. 3. Predict which...
-
Describe the characteristic infrared absorption frequencies that would allow you to distinguish between the following pairs of compounds. (a) 2,3-dimethylbut-2-ene and 2,3-dimethylbut-1-ene (b)...
-
Suppose you invest 52%, 28%, and 20% of your wealth into a stock, the market, and a risk-free asset, respectively. The beta of the stock is 1.1. What is the beta of the portfolio?
-
An investment of $21745, earning compound interest, grows by $2278 in one year. At this rate of growth, how long will it take for the original investment to double?
-
On September 12, 3,400 shares of Denver Company's common stock are acquired at a price of $58 per share plus a $170 brokerage commission. On October 15, an $1.20-per-share dividend was received on...

Study smarter with the SolutionInn App


