Explain why the hydration of this alkene occurs 1015 times faster than the hydration ofethene: OH H20
Question:
Explain why the hydration of this alkene occurs 1015 times faster than the hydration ofethene:
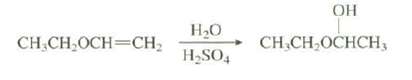
Transcribed Image Text:
OH H20 CH,CH,OCHCH3 CH.CH,OCH=CH, H,SO,
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 63% (22 reviews)
The carbocation intermediate formed from this vinyl eth...View the full answer

Answered By

Grace Igiamoh-Livingwater
I am a qualified statistics lecturer and researcher with an excellent interpersonal writing and communication skills. I have seven years tutoring and lecturing experience in statistics. I am an expert in the use of computer software tools and statistical packages like Microsoft Office Word, Advanced Excel, SQL, Power Point, SPSS, STATA and Epi-Info.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The alkene 3,3-dimethyl-1 -butene undergoes acid-catalyzed hydration with rearrangement. Use the mechanism of hydration and rearrangement to predict the structure of the hydration product of this...
-
Explain why compound A reacts faster than compound B when they undergo solvolysis in aqueous acetone. CH C-Cl CH3 CH CH
-
Why does a heavy parachutist fall faster than a lighter parachutist who wears a parachute of the same size?
-
The membrane filter technique is used to test a polluted water sample for coliform group. Three different dilutions of the water sample were prepared and each was filtered through 5 filter membranes....
-
Discuss the major issues in implementing BI. Discuss.
-
Paul Regan, bookkeeper of Hampton Co., has been up half the night trying to get his trial balance to balance. His results are on page 133. Ken Small, the accountant, compared Pauls amounts in the...
-
What are examples of Human Systems Environment threats and their sources?
-
(Three Differences, Multiple Rates, Future Taxable Income) During 2010, Graham Co.'s first year of operations, the company reports pretax financial income of $250,000. Graham's enacted tax rate is...
-
Police officers noticed two capsules on top of Mr. Rochin's dresser table. They asked Mr. Rochin whose "stuff" is this. Mr. Rochin grabbed the capsules and put them in his mouth and swallowed the...
-
Megatronics Corporation, a massive retailer of electronic products, is organized in four separate divisions. The four divisional managers are evaluated a year-end, and bonuses are awarded based on...
-
Explain why this reaction occurs with anti-Markovnikov regiochemistry: CI + CF,CH,CH CF;CH=CH2 + HCI
-
The addition of HCl to alkynes proceeds through a vinyl cation intermediate. Explain which of the two possible vinyl cations that could be formed from the addition of HCl to propyne is morestable....
-
The following data have been condensed from LA Grande Corporations report of 2012 operations (in millions of euros): 1. Prepare the 2012 income statement in contribution form, ignoring income taxes....
-
1. State any three factors that indicate the continued growth in data warehousing and business intelligence. Can you think of some examples? 2. Why do data warehouses continue to grow in size,...
-
Durkheim noted that psychological explanations are insufficient when analyzing social behavior. In your own words, tell what this statement means?
-
Let w = 2/3 = -1+i3, let R = Z[w]. Let p be a positive == prime integer which is not equal to 3. (a) Prove that R is an Euclidean domain, and find all the units of R. (b) Prove that the ideal pR is a...
-
Apex Energy is evaluating the economics of building a 1000-megawatt nuclear power plant for $1.4 billion. The plant will have net revenue of $400 million per year and will operate for 30 years. At...
-
How do cultural dimensions, as proposed by Hofstede and other cultural theorists, inform our understanding of cross-cultural variations in organizational values, communication styles, and management...
-
Reversible heat engines 1 and 2 are connected to each other in such a way that the output temperature of engine 1 is the input temperature of engine \(2, T_{\text {lout }}=T_{2 \text { in }}\), and...
-
Suppose that fraction used = / 1.0 + 0.1Mt. for some parameter 1. Write the discrete-time dynamical system and solve for the equilibrium. Sketch a graph of the equilibrium as a function of ....
-
For a typical equilibrium problem, the value of K and the initial reaction conditions are given for a specific reaction, and you are asked to calculate the equilibrium concentrations. Many of these...
-
The amino acid threonine, (2S, 3R)-2-amino-3-hydroxybutanoic acid, has two chirality centers. (a) Draw a Fischer projection of threonine. (b) Draw a Fischer projection of a threonine diastereomer,...
-
Hemoglobin has pI = 6.8. Does hemoglobin have a net negative charge or net positive charge at pH = 5.3? At pH = 7.3?
-
Show how you could prepare the following -amino acids from the appropriate carboxylic acids: (a) Phenylalanine (b) Valine
-
The following financial statements are for Microsoft, Inc. FY2023 and FY2022. Calculate the cash flows from operations as it would appear on the FY2023 statement of cash flows. (Hint: include all...
-
Simplify the expression.
-
A 25-year loan of 175,000 is negotiated with the borrower agreeing to repay principal and interest at 7%. A level payment of 10,000 will apply during the first twelve years and a higher level payment...

Study smarter with the SolutionInn App


