Explain why this reaction occurs with anti-Markovnikov regiochemistry: CI + CF,CH,CH CF;CH=CH2 + HCI
Question:
Explain why this reaction occurs with anti-Markovnikov regiochemistry:
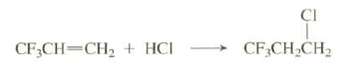
Transcribed Image Text:
CI + CF,CH,CH CF;CH=CH2 + HCI
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (12 reviews)
In this reaction the primary carbocation intermediate is ...View the full answer

Answered By

Sarah Khan
My core expertise are:
-_ Finance
-_ Business
-_ Management
-_ Marketing Management
-_ Financial Management
-_ Corporate Finance
-_ HRM etc...
I have 7+ years of experience as an online tutor. I have hands-on experience in handling:
-_ Academic Papers
-_ Research Paper
-_ Dissertation Paper
-_ Case study analysis
-_ Research Proposals
-_ Business Plan
-_ Complexed financial calculations in excel
-_ Home Work Assistance
-_ PPT
-_ Thesis Paper
-_ Capstone Papers
-_ Essay Writing etc...
5.00+
91+ Reviews
92+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Explain why the reaction shown in eq. 7.19 occurs much more easily than the reaction (That is, why is it necessary to protonate the alcohol before ionization can occur?) (CH) C-OH(CHCHO
-
Explain why hydroxide ion catalyzes the reaction of piperidine with 2,4-dinitroanisole, but has no effect on the reaction of piperidine with 1-chloro-2,4-dinitrobenzene. piperidine
-
Explain why nitration of quinoline (eq. 13.7) occurs mainly at C5 and C8. No 2 (13.7 0C 5-nitroquinoline NO2 8-nitroquinoline
-
MULTIPLE CHOICE: 6. The stage of production at which the individual jointproducts are identified is referred to as the: A. Split-off point B. Joint point C. Separate identification point D. Relative...
-
What is BI governance?
-
An ideal gas follows the three-part process shown in FIGURE 18-29. At the completion of one full cycle, find (a) The net work done by the system, (b) The net change in internal energy of the system,...
-
A stock price is governed by geometric Brownian motion with \(\mu=.20\) and \(\sigma=.40\). The initial price is \(S(0)=1\). Evaluate the four quantities E[In S(1)], E[S(1)], stdev[In S(1)]...
-
1. A price-discriminating firm will charge a higher price to consumers with a relatively ________ (elastic/ inelastic) demand and a lower price to consumers with a relatively ________...
-
Compute ROA, ROE and ROFL and Interpret the Effects of Leverage Basic income statement and balance sheet information is given below for six different cases. For each case, the assets are financed...
-
Refer to the Hungry Dawg Restaurant example presented in this chapter. Health claim costs actually tend to be seasonal, with higher levels of claims occurring during the summer months (when kids are...
-
Explain the difference in the percentages of the products in these two hydroborationreactions: CH3 CH, QH CH, 1) BH3. THF 2) H,O,, NAOH CH,CH-CHCH-CH; + CH;CHCH,CHCH3 (43%) CH,CHCH=CHCH, (57%) 1)...
-
Explain why the hydration of this alkene occurs 1015 times faster than the hydration ofethene: OH H20 CH,CH,OCHCH3 CH.CH,OCH=CH, H,SO,
-
Find the area of the figure described. Round your answer to two decimal places. A regular pentagon with radius 10.6 inches.
-
We have a closed system containing water at a pressure of 0.2 bar. The specific volume is 5,427.19 cm/g. For this vapor-liquid mixture, report the fraction of vapor. Use the steam tables and report...
-
Q3. Consider an angular momentum 1 system, represented by the state vector 14) = 1 1 4 26 -3 4.4 (868) 1 = 1 2 10 What is the probability that a measurement of 1x yields the value 0 ? 6/8 Can you...
-
Questions 1-2 The switch of the circuit shown in the figure is closed at time t = 0. The capacitor is initially = 12 V, R = 50, R: uncharged and the battery has no internal resistance. E = C = 100...
-
Questions 3-4 A rectangular wire loop carrying current of I = 200 mA has a side of a = 2 cm and b - 6 cm as shown in the figure. The loop is placed to a uniform magnetic field of B= 2T. Initially,...
-
A gardener is using a tool as shown in the figure to pull weeds. 6.0 cm 24 cm You will notice that the tool is designed with a built-in pivot point about which the torques are applied. If the...
-
Describe the liquidity preference theory.
-
Solve for the equilibria of the following discrete-time dynamical systems Pr pt+1 = Pr+2.0(I-Pr)
-
Write the equilibrium expression (K) for each of the following gas-phase reactions. a. N(g) + O(g)=2NO(g) b. NO(g)2NO(g) c. SiH4(g) + 2Cl(g) d. 2PBr3(g) + 3Cl(g) SiCl4(g) + 2H(g) 2PC13(g) + 3Br(g)
-
One of the steps in the biological pathway for carbohydrate metabolism is the conversion of fructose 1, 6-bisphosphatc into dihydroxyacetone phosphate and glyceraldehydes 3-phosphate. Propose a...
-
L-Fucose, one of the eight essential monosaccharide?s, is biosynthesized from GDP-D-mannose by the following three-step reaction sequence, where (H)P?guano sine diphosphate (a ribonucleoside...
-
Of the 19 L amino acids, 18 have the S configuration at the carbon. Cysteine is the only L amino acid that has an R configuration. Explain.
-
Discuss the dynamic organization of the cytoskeleton and its pivotal role in cellular motility, intracellular transport, and structural integrity .
-
Would you support the idea of a government issued Digital currency? Why ? and why not?
-
To protect her savings against further inflation and to help her prepare for a healthy financial future, Hanna Lind deposits $9,100 in an investment account earning 6% interest compounded quarterly....

Study smarter with the SolutionInn App


