Explain why the proto-nation of an amide occurs at the O rather than the N. 0: CHCNH,
Question:
Explain why the proto-nation of an amide occurs at the O rather than the N.
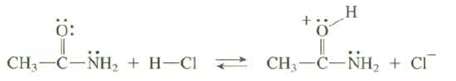
Transcribed Image Text:
0: CH–C−NH, + H—C < * H CH,—C−NH, + CF
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (15 reviews)
The unshared electron pair on the nitrogen of this compound is in a ...View the full answer

Answered By

ANDREW KIPRUTO
Academic Writing Expert
I have over 7 years of research and application experience. I am trained and licensed to provide expertise in IT information, computer sciences related topics and other units like chemistry, Business, law, biology, biochemistry, and genetics. I'm a network and IT admin with +8 years of experience in all kind of environments.
I can help you in the following areas:
Networking
- Ethernet, Wireless Airmax and 802.11, fiber networks on GPON/GEPON and WDM
- Protocols and IP Services: VLANs, LACP, ACLs, VPNs, OSPF, BGP, RADIUS, PPPoE, DNS, Proxies, SNMP
- Vendors: MikroTik, Ubiquiti, Cisco, Juniper, HP, Dell, DrayTek, SMC, Zyxel, Furukawa Electric, and many more
- Monitoring Systems: PRTG, Zabbix, Whatsup Gold, TheDude, RRDtoo
Always available for new projects! Contact me for any inquiries
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Proto Chemical produces and sells an ice-melting granular used on roadways and sidewalks in winter. The company annually produces and sells about 300,000 lbs of its granular. In its ten-year history,...
-
Explain why sodium amide (NaNH2) and phenyl lithium (C6H5Li) are leveled to the same base strength in aqueous solution. Write the chemical reactions that occur when these reagents are added to water.
-
Explain why sodium amide cannot be used to form a carbanion from an alkane in a reaction that favors products?
-
You have $300,000 saved for retirement. Your account earns 6% interest. How much will you be able to pull out each month, if you want to be able to take withdrawals for 20 years? $ Get help: Video
-
Explain the difference between the traditional direct labour rate variance and a pure rate variance?
-
The Veblen Company and the Knight Company are identical in every respect except that Veblen is not levered. Financial information for the two firms appears in the following table. All earnings...
-
Provide examples of Web services and discuss the contribution of Web services to the efficiency of information systems.
-
Horn Company accumulates the following adjustment data at December 31. Indicate (a) The type of adjustment (prepaid expense, accrued revenues and so on), and (b) The status of accounts before...
-
Grossnickle Corporation issued 20-year, noncallable, 9.7% annual coupon bonds at their par value of $1,000 one year ago. Today, the market interest rate on these bonds is 7.4%. What is the current...
-
Draft a quarterly cash budget for the Zoo Extravaganza (ZE) for the first two quarters of the coming year. Use just the information in this problem. 1. Interest is paid on the last day of the year...
-
Explain which of these compounds is the weaker base? :Z 0 HIN:
-
Amino acids contain both a basic functional group, the amine, and an acidic functional group, the carboxylic acid. Thus, they can undergo an internal acid-base re-action as shown in the following...
-
In 20X0, Marquette County opened a landfill that was expected to accept waste for four years. The following table indicates the estimates county officials made at the end of each of the four years:...
-
Eastern European farmers produce a variety of fruit juices, which they wish to dehydrate to prolong shelf-life and facilitate transportation. One very simple dehydration method is to put the juice in...
-
In the benzene adsorber of Example 9.6, the flow rate is increased to \(0.25 \mathrm{~m}^{3} / \mathrm{s}\). Calculate the breakthrough time and the fraction of the bed adsorption capacity that has...
-
It is desired to remove \(90 \%\) of the sulfur dioxide in a flue gas stream at \(298 \mathrm{~K}\) and \(1 \mathrm{~atm}\) by countercurrent absorption with pure water at the same temperature, using...
-
It is desired to reduce the ammonia content of \(0.05 \mathrm{~m}^{3} / \mathrm{s}\) of an ammonia-air mixture ( \(300 \mathrm{~K}\) and \(1 \mathrm{~atm}\) ) from \(5.0 \%\) to \(0.04 \%\) by volume...
-
Germanium tetrachloride \(\left(\mathrm{GeCl}_{4} ight)\) and silicon tetrachloride \(\left(\mathrm{SiCl}_{4} ight)\) are used in the production of optical fibers. Both chlorides are oxidized at high...
-
It was 1983 when Bernard J. (aka "Bernie") Ebbers founded Long Distance Discount Service (LDDS), a discount long-distance telephone company, \({ }^{148}\) Local legend has it that Mr. Ebbers, a...
-
The Adjusted Trial Balance columns of a 10-column work sheet for Webber Co. follow. Complete the work sheet by extending the account balances into the appropriate financial statement columns and by...
-
Prove formula (b) of Theorem 3. Data from Formula b of theorem 3 M (b) c = ne = i=1
-
Draw structures corresponding to the following IUPAC names: (a) 2, 3-Dimethylhexanoic acid (b) 4-Methylpentanoic acid (c) Trans-1, 2-Cyclobutanedicarboxylic acid (d) o-Hydroxybenzoic acid (e) (9Z,...
-
Assume you have a mixture of naphthalene and benzoic acid that you want to separate. How might you take advantage of the acidity of one component in the mixture to effect a separation?
-
The Ka for dichloroacetic acid is 3.32 x 102. Approximately what percentage of the acid is dissociated in a 0.10M aqueous solution?
-
Suppose that the net income view were to be correct in Problem 1. Describe a profitable investment strategy for investors in the companys bonds and equity if the company were to reduce its debt. What...
-
A piece of land in Ottawa with an area of 0.5 square kilometers is priced at 5200 Canadian dollars. If there are 0.9955 Canadian dollars per (U.S.) dollar, then what is the price in dollars per...
-
Marc and Mikkel are married and file a joint tax return. Marc and Mikkel earned salaries this year of $ 6 6 , 2 0 0 and $ 2 5 , 2 0 0 , respectively. In addition to their salaries, they received...

Study smarter with the SolutionInn App


