Explain which of these compounds is the weaker base? :Z 0 HIN:
Question:
Explain which of these compounds is the weaker base?
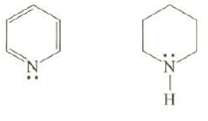
Transcribed Image Text:
:Z 0 HIN:
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 73% (15 reviews)
The basic electrons are on a nitrogen atom in both ...View the full answer

Answered By

Bhartendu Goyal
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions. I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and help them achieve great subject knowledge. I have expertise in computer subjects like C++, C, Java, and Python programming and other computer Science related fields. Many of my student's parents message me that your lessons improved their children's grades and this is the best only thing you want as a tea...
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Explain whether each of these compounds is chiral or not: a) d) H C H c=c=c CHCH CCH HC PhwSi-OCH-CH b) CHCH Ph-N-CHCH, HC Br H C CH30 CH CO H c) f) H H HC CH30 CCH CH3 COH CH3 CO H
-
Explain whether each of these compounds is chiral or not: , CCH3 C=C=C] CH3 . a) b) C=C=C, , , . c) CH;O. `CH3 - . d) CH;O CO,H CH;OS Br CH;CH2 e) f) N-CH,CH3 Ph CH;CH2
-
Which of these compounds can be a member of an isomer pair? In each case where isomerism is possible, identify the type or types of isomerism. [Sections 24.2, 24.4] CH2 C C-OH --O NHE Cl (b) CH3CH2CH...
-
when taking a lead role for supplier selection can often help Multiple select question. establish supplier quality standards. determine price. develop the engineering specifications. set conditions...
-
Tahmar Valley Chemical Company manufactures industrial chemicals. The company plans to introduce a new chemical solution and needs to develop a standard product cost. The new chemical solution is...
-
A stock has an expected return of 13.5 percent, its beta is 1.17, and the risk-free rate is 5.5 percent. What must the expected return on the market be?
-
Compare and explain vertical and horizontal layers in application architecture.
-
Mary Givens and Peggy Moser are partners engaged in operating The G&M Doll Shop, which has employed the following persons since the beginning of the year: T. Binn (general office...
-
Suppose the exchange rate between U.S. dollars and Swiss francs is CHF 0.92 = $1.00, and the exchange rate between the U.S. dollar and the euro is $1.00 = 0.84 euro. What is the cross rate of Swiss...
-
Which life cycle model would you follow for developing software for each of the following applications? Mention the reasons behind your choice of a particular life cycle model. Clearly Justify your...
-
Which compound is behaving as the Lewis acid and which as the Lewis base in this reaction? AICI3 T CHCHCHCH3 + AICI CHCHCHCH3
-
Explain why the proto-nation of an amide occurs at the O rather than the N. 0: CHCNH, + HC < * H CH,CNH, + CF
-
Buying a Car: Create a Decision Matrix David, an outrigger canoe racer, needs to buy a new car. He wants a vehicle that will carry his disassembled boat and outrigger. At the same time he will need...
-
Modify the Mathcad program developed in Problem 3.16 to estimate the minimum gas flow rate in strippers so that it can be used to estimate the minimum air flow required for water cooling. Test your...
-
A cooling tower cools \(227 \mathrm{~m}^{3} / \mathrm{h}\) of water from \(314 \mathrm{~K}\) to \(306 \mathrm{~K}\) using a countercurrent forced draft of air entering at \(317 \mathrm{~K}\) and...
-
Electricity prices vary widely from state to state. In Connecticut, for instance, electricity costs \($0.209/kWh,\) while in Illinois the cost is \($0.125/kWh.\) On average, residents of both states...
-
Repeat Example 9.3, but with a feed pressure of 5.0 MPa. Compare your results to those obtained at a feed pressure of \(1.0 \mathrm{MPa}\). Data From Example 9.3:- Consider the air separation problem...
-
A recently installed induced-draft cooling tower (with the fan located at the top) was guaranteed by the manufacturer to cool \(450 \mathrm{~m}^{3} / \mathrm{h}\) of water from \(316 \mathrm{~K}\) to...
-
This section of SOX established a quasi-governmental entity called the Public Company Accounting Oversight Board (PCAOB, but called "Peek-a-Boo") under the direction of the SEC to (1) oversee the...
-
Compile data on consumption and expenditures for the following categories in 30 different countries: (1) food and beverages, (2) clothing and footwear, (3) housing and home operations, (4) household...
-
Sketch the graph of the equation. |y| = 1
-
When crystals of pure ?-glucose are dissolved in water, isomerization slowly occurs to produce ?-glucose. Propose a mechanism for the isomerization. CH2 CH2 - - B-Glucose a-Glucose
-
When glucose (Problem) is treated with NaBH4, reaction occurs to yield sorbitol, a polyalcohol commonly used as a food additive. Show how this reductionoccurs. CH- NABHA -2 Sorbitol Glucose
-
Give IUPAC names for the followingcompounds: (a) Br (b) (c) CH CH3CH2CHCH2CH2CH3 CHH-CH2Co CHCH2C (f) O2- (d) CH CN (e) CHCCH2CHH "Co2 CH2CH2COH
-
Match the statements to the protocols * faster more reliable used for sending data TCP UDP both
-
1. Should managers try to maximize market value of equity or to maximize the accounting value of equity in the balance sheet? Why? 2. What is your understanding of the relationship between risk and...
-
How much risk is appropriate for a government-run annuity system? Is there an appropriate risk-return calculation to be made? Is Social Security risk free? What about political risk?

Study smarter with the SolutionInn App


