Explain why this secondary alcohol reacts with HCl and ZnCl2 in H2O at about the same rate
Question:
Explain why this secondary alcohol reacts with HCl and ZnCl2 in H2O at about the same rate as a primary alcohol (see Problem 8.38)
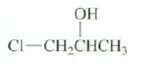
Transcribed Image Text:
OH Cl-CH,CHCH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (11 reviews)
The carbocation which is formed at t...View the full answer

Answered By

Bhartendu Goyal
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions. I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and help them achieve great subject knowledge. I have expertise in computer subjects like C++, C, Java, and Python programming and other computer Science related fields. Many of my student's parents message me that your lessons improved their children's grades and this is the best only thing you want as a tea...
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
A 6.11-g sample of a Cu-Zn alloy reacts with HCl acid to produce hydrogen gas. If the hydrogen gas has a volume of 1.26 L at 22C and 728 mmHg, what is the percent of Zn in the alloy? (Cu does not...
-
Explain why maleic anhydride reacts rapidly with 1,3-butadiene but does not react at all with ethene under thermal conditions. maleic anhydride
-
Explain relationships between primary and secondary boycotts.
-
The task in this design project is to design an actuation system to power the ram's reciprocating motion in a small-size shaper. The power source of the actuation system is an AC motor with 0.75 hp...
-
In each case below, fill in the missing amount: 20 000 kg Work in process, 1 April 1 Units started during April 32 000 kg Units completed during April 6000 kg Work in process, 30 April Work in...
-
Eve and Tom own 40% and 60%, respectively, of the ET Partnership, which manufactures clocks. The partnership is a limited partnership, and Eve is the only general partner. She works full-time in the...
-
Air enters a 0.5 -ft inside diameter duct with \(p_{1}=20\) psia, \(T_{1}=80^{\circ} \mathrm{F}\), and \(V_{1}=200 \mathrm{ft} / \mathrm{s}\). What frictionless heat addition rate in Btu/s is...
-
The financial statements of Lioi Steel Fabricators are shown below both the actual results for 2010 and the projections for 2011. Free cash flow is expected to grow at a 6% rate after 2011. The...
-
Use the information below and prepare the Multi-Step Income Statement for Hot Place on August 31. Sales $1,250,000 Interest Revenue 25 Sales Discounts 15,000 Sales Returns and Allowances 30,000 Cost...
-
Which of the graphs in Fig. Q25.12 best illustrates the current I in a real resistor as a function of the potential difference V across it? Explain. Figure Q25.12 (a) (b) (c) (d)
-
The Lucas test is used to check for the presence of an alcohol functional group in an unknown compound. The test reaction is shown in the following equation: Smaller alcohols are soluble in the...
-
What reagent and solvent would you use to carry out the following transformations? -Cl -Br Ph. b) C,CH Ph CH CH OCCH3 Br Ph. Ph-C-OCH (Racemic) c) CH,CH CH-CH3 -NH,CH, C d) tom Br e) a)
-
Refer to Exercise 8.54. Find the amount of sleep that is exceeded by only 25% of students. In exercise 8.54 University and college students average 7.2 hours of sleep per night, with a standard...
-
Records show that the probability is 0.00008 that a truck will have an accident on a certain highway. Use the formula for the Poisson distribution to approximate the probability that at least 5 of...
-
The daily number of orders filled by the parts department of a repair shop is a random variable with \(\mu=142\) and \(\sigma=12\). According to Chebyshev's theorem, with what probability can we...
-
Nurse Practitioner Nancy Masters broke up with her control freak boyfriend, Joe Jerque, after a three-year relationship that was going nowhere but down. Despite her repeated pleas for counseling, he...
-
Show that the least-squares estimate of \(\boldsymbol{\beta}\) (say \(\hat{\boldsymbol{\beta}}_{(i)}\) ) with the \(i\) th observation deleted can be written in terms of the estimate based on all...
-
Why have developing countries such as China and India become important international markets?
-
(a) Determine the critical values for a right-tailed test of a standard population deviation with 15 degrees of freedom at \(\alpha=0.1\) level of significance. (b) Determine the critical values for...
-
When an electric field is applied to a shallow bath of vegetable oil, why do tiny bits of thread floating in the oil align with the field like compasses in a magnetic field?
-
Evaluate each using the values given. (4x (y + y)); use x = -4, and y=-
-
How would you prepare the following compound using a Michaelreaction?
-
What products would result after hydrolysis from reaction of the enamine prepared from cyclopentanone and pyrrolidine with the following , -unsaturated acceptors? (a) CH 2 =CHCO 2 Et (b) H 2 C=CHCHO...
-
Show how you might use an enamine reaction to prepare each of the followingcompounds: (b) (a) CH2CH2CO2CH3 CH2CH2CN
-
You are the new controller for Moonlight Bay Resorts. The company CFO has asked you to determine the company's interest expense for the year ended December 31, 2021. Your accounting group provided...
-
How do the dynamics change in Calm Down by Selena Gomez ?
-
A company's inventory records report the following: August 1 August 5 August 12 Beginning balance Purchase Purchase 23 units @ $13 18 units $12 22 units @ $13 On August 15, it sold 46 units. Using...

Study smarter with the SolutionInn App


