Show how you might use an enamine reaction to prepare each of the followingcompounds: (b) (a) CH2CH2CO2CH3
Question:
Show how you might use an enamine reaction to prepare each of the followingcompounds:
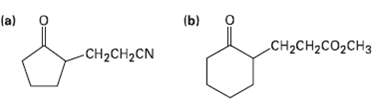
Transcribed Image Text:
(b) (a) CH2CH2CO2CH3 CH2CH2CN
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 44% (9 reviews)
Analyze the product for the Michael acceptor and the ...View the full answer

Answered By

Robert Kiptanui
I am persiuing my B tech from Maulana Azad National Institute of Technology. I am teaching student from past 3 years . I am a part of an NGO named "AAROHA" this is a firm which
give education to financially unstable students.my hobbies are problem solving and article writing. Problem-solving is an essential skill that is valuable in many areas of life, whether it be in academics, business, or personal endeavors. It involves using critical thinking and logical reasoning to find solutions to complex issues. Article writing, on the other hand, is a great way to communicate your ideas and thoughts to a wide audience. It allows you to share your knowledge and expertise on a particular topic and to engage with others who have similar interests.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Show how you might use thioacetal formation and Raney nickel desulfurization to convert: (a) cyclohexanone to cyclohexane and (b) benzaldehyde to toluene.
-
Show how you might use a nucleophilic substitution reaction of 1-bromopropane to synthesize each of the following compounds. (You may use any other compounds that are necessary.) (a) (b)...
-
Using a Stork enamine synthesis, show how you might accomplish each of the following transformations. (a) (b) (c) H
-
Strickland Co. currently charges manufacturing over-head costs to products using machine hours. However, company management believes that the use of ABC would provide more realistic cost estimates...
-
Is it possible to organize too much to meet the needs of the environment? This would be a case of strategic misfit. How would you know if a misfit has occurred? Think of an example of misfit caused...
-
(a) Calculate the standard enthalpy of formation of gaseous diborane (B2H6) using the following thermochemical information: (b) Pentaborane (B5H9) is another boron hydride. What experiment or...
-
How would you explain the quantum mechanics concept of superposition to a friend?
-
1. What problems such as lawsuits, reputation, and public image would GarageTek face if they closed failing franchises? 2. Is shuttering the failed franchises the right move for Shuman? What are his...
-
What are the pros and cons to automate employment benefits tasks and functions?
-
? ? Preparing financial statements and closing entries Paulson Corporation Adjusted Trial Balance December 31, 20- DERIT CREDIT ACCOUNT TTLE Cash 28 3 6 211 Petty Cash 1200 Accounts Receivable...
-
What products would result after hydrolysis from reaction of the enamine prepared from cyclopentanone and pyrrolidine with the following , -unsaturated acceptors? (a) CH 2 =CHCO 2 Et (b) H 2 C=CHCHO...
-
What product would you expect from a Robinson annulations reaction of 2-methyl-1, 3-cyclopentanedione with3-buten-2-one? -CH + H%3DH 2-Methyl-1,3-cyclo- pentanedione 3-Buten-2-one
-
What statistical procedure can be used to test whether there is a significant difference in the median gray levels between cataractous and normal eyes? Ophthalmology A camera has been developed to...
-
In your own words, answer this unit's discussion questions in a main post (recommended minimum 200 words). Assignment Details Should people just say "live and let live" when they look at the...
-
1. Consider the Cobb-Douglas production function where real aggregate output Y is given by Y=AK (LXE)-a, in which A-60 is a parameter measuring the productivity of the available technology, K is the...
-
The North American Transportation Company (NATC) is a very large organization that provides continent-wide facilities for the shipping of goods, from tonnes of wheat and iron ore to individual...
-
Written Assignment: Answer each below using correct grammar, punctuation and complete sentences. 1. Identify and discuss four basic steps in a risk management program. 2. Define concepts of...
-
A company currently has $25 million in excess cash and no debt. The company expects to generate additional net after-tax cash flows of $20 million per year in subsequent years and will pay out these...
-
What is the rotational speed of a watch's second hand? Its hour hand?
-
Chao, Louis, and Mari, unrelated individuals, own all of the shares of Cerise Corporation. All three shareholders have been active in the management of Cerise since its inception. In the current...
-
Ethanol is a possible fuel. Use average bond energies to calculate H rxn for the combustion of ethanol. CH3CHOH(g) + 3 O(g) 2 CO(g) + 3 HO(g)
-
(a) Give two syntheses for (CH3)2CH - O - CH2CH3, and explain which synthesis is better. (b) A student wanted to synthesize methyl tert-butyl ether, CH3 - O - C (CH3)3. He attempted the synthesis by...
-
When ethyl bromide is added to potassium tert-butoxide, the product is ethyl tert-butyl ether. (a) What happens to the reaction rate if the concentration of ethyl bromide is doubled? (b) What happens...
-
When tert-butyl bromide is heated with an equal amount of ethanol in an inert solvent, one of the products is ethyl tert-butyl ether. (a) What happens to the reaction rate if the concentration of...
-
1. What would a graph of the speed of an object that moves at a constant speed look like? (hint: curved line, broken line, etc.) 2. A car travels 120 km in 1250 seconds. What is the speed in...
-
Today we are going to calculate the electric field of two concentric non-conducting charged cylinders (separated by a neutral insulator) of infinite length in all regions of space. We will be using...
-
You are Professional Accountant ( SA ) for registered accountants. A client of yours in the SME sector is speculating shares on listed company on the JSE. The detail and name of the listed company is...

Study smarter with the SolutionInn App


