Figure gives the energy levels for an electron trapped in a finite potential energy well 450eV deep.
Question:
Figure gives the energy levels for an electron trapped in a finite potential energy well 450eV deep. If the electron is in the n = 3 state, what is its kineticenergy?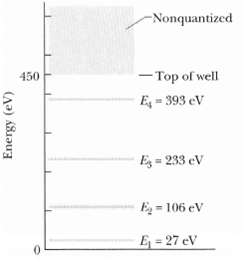
Transcribed Image Text:
-Nonquantized - Top of well 450 E 393 eV E = 233 eV Eg = 106 eV E=27 eV Energy (eV)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 55% (9 reviews)
From figure we see that the sum of the kinetic and potential energies in that p...View the full answer

Answered By

Charles mwangi
I am a postgraduate in chemistry (Industrial chemistry with management),with writing experience for more than 3 years.I have specialized in content development,questions,term papers and assignments.Majoring in chemistry,information science,management,human resource management,accounting,business law,marketing,psychology,excl expert ,education and engineering.I have tutored in other different platforms where my DNA includes three key aspects i.e,quality papers,timely and free from any academic malpractices.I frequently engage clients in each and every step to ensure quality service delivery.This is to ensure sustainability of the tutoring aspects as well as the credibility of the platform.
4.30+
2+ Reviews
10+ Question Solved
Related Book For 

Probability & Statistics For Engineers & Scientists
ISBN: 9780130415295
7th Edition
Authors: Ronald E. Walpole, Raymond H. Myers, Sharon L. Myers, Keying
Question Posted:
Students also viewed these Modern Physics questions
-
The penetration distance ? in a finite potential well is the distance at which the wave function has decreased to 1/e of the wave function at the classical turning point: The penetration distance can...
-
Discuss what happens to the energy levels for an electron trapped in a one-dimensional box as the length of the box increases.
-
Suppose that an electron trapped in a one-dimensional infinite well of width 250 pm is excited from its first excited state to its third excited state. (a) What energy must be transferred to the...
-
Toby dies on 2 March 2021, leaving an estate valued at 400,000. None of the transfers made on death are exempt from IHT. Calculate the IHT due on the estate if the total of the gross chargeable...
-
Talk to a financial planner or an insurance agent about the financial difficulties faced by people who lack adequate home and auto insurance. What common coverages do many people overlook?
-
Express the internal shear and moment in the cantilevered beam as a function of x and then draw the shear and moment diagrams. 300 Ib 200 lb/ft 6 ft
-
The number of weekly breakdowns of a computer is a random variable having a Poisson distribution with \(\lambda=0.2\). What is the probability that the computer will operate without a breakdown for 3...
-
What are the likely reasons and rationalizations Matt will give for making the payments discussed in the case?
-
There are only 4 teams in a league. The winning percentages of the teams are given as follows: Team A: 58%, Team B: 30%, Team C: 62% and Team D: 82%. Calculate the standard deviation.
-
The area of cost and management accounting is ever changing and regularly evolving due to ever growing changes in the business scenario and financial markets. With the ever evolving accounting...
-
An electron in the n = 2 state in the finite potential well of figure absorbs 400eV of energy from an external source. Using the energy-level diagram of figure, determine the electron's kinetic...
-
(a) Show that for the region x > L in the finite potential well of figure ?(x) = De2ks is a solution of Schrodinger's equation in its one-dimensional form, where D is a constant and k is positive.(b)...
-
The structure of ethylene is \(\mathrm{H}_{2} \mathrm{C}=\mathrm{CH}_{2}\). Estimate the enthalpy of reaction when \(1 \mathrm{kmol}\) of ethylene is completely oxidised. Compare the value obtained...
-
You decide that it may be appropriate to offer Gillian some feedback about her behavior. When you ask her if she would like some feedback, she says yes. 1. What type of question are you using when...
-
Since you were not present during the exchange between Paul and Gillian, you are not aware of what happened. You are reluctant to give Gillian any advice. 1. Mention an "I feel" message that you...
-
For this task you must research each of the following topics, and provide a report that identifies your understanding of each topic. What is the Universal Declaration of Human Rights and how does it...
-
Select a commercial, film, or TV program (any media). What is the main message being conveyed? Identify any bias or stereotypes through media representation (e.g., gender, race, class, ability, etc.)...
-
Explain how what training must managers be receiving to handle cases of discrimination at the workplace? You have just moved to Canada. The new community, new traditions, and habits it is not an easy...
-
You have a hand in 5-card poker with 2 kings, 1 ace, and 2 numbered cards. You consider trading in the two numbered cards and the ace for three new cards. What is the probability that at least one of...
-
Consider a game of poker being played with a standard 52-card deck (four suits, each of which has 13 different denominations of cards). At a certain point in the game, six cards have been exposed. Of...
-
In which of the following molecules would you expect to find delocalized molecular orbitals? Explain. (a) C 2 H 4 ; (b) SO 2 ; (c) H 2 CO.
-
The following is the distribution of the readings obtained with a Geiger counter of the number of particles emitted by a radioactive substance in 100 successive 40-second intervals: (a) Verify that...
-
The following are the hours of operation to failure of 38 light bulbs. Use a suitable statistical computer program to test whether the mean failure time of such light bulbs is significantly less than...
-
The following are the hours of operation to failure of 38 light bulbs. Use a suitable statistical computer program to test whether the mean failure time of such light bulbs is significantly less than...
-
ENG 571, Theory of Energy and Sustainability Engineering, Sp 2023 HW# 3, Due on Friday, April 28 (*two days later than stated on the syllabus) Note: Students who are tasking this course for 4 hours...
-
Discuss your personal experience with dynamic pricing ( airlines , hotels, Uber, event tickets, etc. ) . Also, discuss any professional experience including: What revenue - management methods does...
-
This assignment requires you to play the role of an international marketing manager from an international consulting company. Your mission is helping your selected Japanese company in Japan to...

Study smarter with the SolutionInn App


