Figure shows an x-ray diffraction pattern for a-iron taken using a diffract meter and monochromatic x-radiation having
Question:
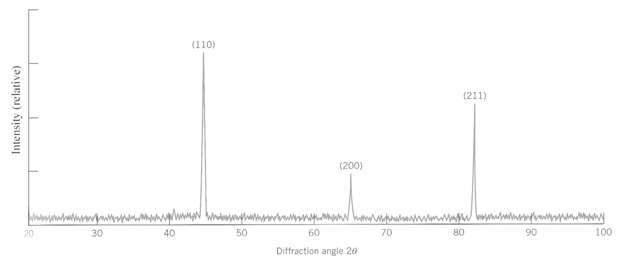
Figure shows an x-ray diffraction pattern for a-iron taken using a diffract meter and monochromatic x-radiation having a wavelength of 0.1542 nm; each diffraction peak on the pattern has been indexed. Compute the interplanar spacing for each set of planes indexed; also determine the lattice parameter of Fe for each of thepeaks.
Transcribed Image Text:
(110) (211) (200) 90 100 20 30 40 50 60 70 OR Diffraction angle 20 Intensity (relative)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 46% (13 reviews)
For each peak in order to compute the interplanar spacing and the lattice parameter we must employ ...View the full answer

Answered By

Vikash Gupta
I am graduated in Physics in 2018, from KIRORIMAL COLLEGE, University of Delhi. Now I am persuing Master's degree in physics. I like to do physics problems. I have experience of 1 year in tutoring. I think Physics is the only subject where you understand things,how they are happening . In physics you learn Maths and apply it. So I would like to join your platform to solve many Physics problems.
5.00+
5+ Reviews
10+ Question Solved
Related Book For 

Fundamentals of Materials Science and Engineering An Integrated Approach
ISBN: 978-1118061602
4th Edition
Authors: David G. Rethwisch
Question Posted:
Students also viewed these Materials Science Engineering questions
-
UV radiation having a wavelength of 54 nm falls on gold metal, to which electrons are bound by 4.82 eV. What is the maximum velocity of the ejected photoelectrons?
-
Figure 3.22 shows an x-ray diffraction pattern for α-iron taken using a diffractometer and monochromatic x-radiation having a wavelength of 0.1542 nm; each diffraction peak on the...
-
A light ray of wavelength 589 nm is incident at an angle θ on the top surface of a block of polystyrene surrounded by air, as shown in figure. (a) Find the maximum value of...
-
Sally works as a welder at Heavy Iron Works Company. She is the only female welder in a shop with ten male welders. Teddy is the manager of that shop with Sally and the ten male welders. Sally is...
-
Discuss which teapot holds more liquid, and why.
-
What role did organizational ethics play in the Beazer Homes fraud? Is this something the auditors of Deloitte should have been more conscious of? Explain.
-
Look through several professional society magazines such as Civil Engineering, Mechanical Engineering, or ASEE Prism. Count the advertisements for CFD software and calculate an "ad density" in...
-
Paul Golding and his wife, Nancy, established Crunchy Chips in 1938. (Nancy sold her piano to help raise capital to start the business.) Paul assumed responsibility for buying potatoes and selling...
-
A metallic sphere of diameter 5 cm is initially at a uniform temperature of 300oC. Suddenly the metal sphere is immersed in a fluid at 30oC. The convective heat transfer coefficient between the...
-
Pacifico Company, a U.S.-based Importer of beer and wine, purchased 1,500 cases of Oktoberfest-style beer from a German supplier for 375,000 euros. Relevant U.S. dollar exchange rates for the euro...
-
For which set of crystallographic planes will a first-order diffraction peak occur at a diffraction angle of 46.21( for BCC iron when monochromatic radiation having a wavelength of 0.0711 nm is used?
-
The diffraction peaks shown in figure are indexed according to the reflection rules for BCC (i.e., the sum h + k + l must be even). Cite the h, k, and l indices for the first four diffraction peaks...
-
Why is the payroll system of most entities computerized?
-
Evaluate of 2 +4 C 2-3 dz where where c y 12)=5.
-
Mrs . Perry's total income consisted of $ 1 0 , 0 0 0 in eligible dividends received from taxable Canadian corporations. Mrs . Perry's BPA and dividend tax credits are sufficient to offset all of her...
-
Refer to the following bank reconciliation: Bank Book Balance, June 3 0 , 2 0 2 5 $ 1 1 , 2 4 0 Balance, June 3 0 , 2 0 2 5 $ 1 0 , 2 0 0 Add: Add: Deposit in transit 3 , 1 1 0 Note collected by bank...
-
Discuss how the market mechanism works in a free market economy and how a shift in the supply and/or demand curve is different from a change in the quantity demanded or supplied. How does the free...
-
1. What is a common defense (sometimes successful and sometimes unsuccessful) to an allegation of employment related discrimination? Explain 2. Before terminating an employee for just cause {based on...
-
Consider a. Using the final-value theorem, if applicable, evaluate x s s x s s . b. Confirm the result of (a) by evaluating lim t x ( t ) lim t x ( t ) . X(s) = s+3s+1 s(s+1)2
-
A company pledges their receivables so they may Multiple Choice Charge a factoring fee. Increase sales. Recognize a sale. Collect a pledge fee. Borrow money. Failure by a promissory notes' maker to...
-
A hot metal object is submerged in cold water. The rate at which the object cools (in degrees per minute) is a function (t) of time. Which quantity is represented by the integral S f(t) dt?
-
It is necessary to select a metal alloy for an application that requires a yield strength of at least 345 MPa (50,000 psi) while maintaining a minimum ductility (%EL) of 20%. If the metal may be cold...
-
A cylindrical rod of 1040 steel originally 15.2 mm (0.60 in.) in diameter is to be cold worked by drawing; the circular cross section will be maintained during deformation. A cold-worked tensile...
-
A cylindrical rod of copper originally 16.0 mm (0.625 in.) in diameter is to be cold worked by drawing; the circular cross section will be maintained during deformation. A cold-worked yield strength...
-
3. Create a chart for gross margin less R&D and capital costs calculations. a. Calculate gross margin less R&D and capital costs for options A and B. b. Graph total gross margin less R&D and capital...
-
Why was Thomas Paine's "common sense" article significant to the american revolution?
-
Aramex, Inc is currently offering a bond with a par value of $1,000, which is priced at $1,488 in the market. The bond offers a fixed annual coupon rate of 14.05 percent, which is paid semiannually....

Study smarter with the SolutionInn App


