Figure shows a reversible cycle through which 1.00 mol of a monatomic ideal gas is taken. Volume
Question:
Figure shows a reversible cycle through which 1.00 mol of a monatomic ideal gas is taken. Volume Vc = 8.00%. Process bc is an adiabatic expansion, with pb = 10.0 atm and Vb = 1.00 x 10-3 m3. For the cycle, find
(a) The energy added to the gas as heat,
(b) The energy leaving the gas as heat,
(c) The net work done by the gas, and
(d) The efficiency of the cycle.
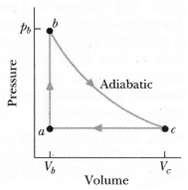
Transcribed Image Text:
Adiabatic V, V. Volume Pressure
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 41% (12 reviews)
a Energy is added as heat during the portion of the process from a to b This portion occurs ...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Fundamentals of Physics
ISBN: 978-0471758013
8th Extended edition
Authors: Jearl Walker, Halliday Resnick
Question Posted:
Students also viewed these Thermodynamics questions
-
A 1.00-mol sample of a monatomic ideal gas is taken through the cycle shown in Figure P22.65. At point A, the pressure, volume, and temperature are Pi , Vi , and Ti , respectively. In terms of R and...
-
Suppose 1.00 mol of a monatomic ideal gas is taken from initial pressure p1 and volume V1 through two steps: (1) an isothermal expansion to volume 2.00V1 and (2) a pressure increase to 2.009 at...
-
The thermodynamic energy of a monatomic ideal gas is given by Find the partial derivatives and write the expression for dU using T ,V, and n as independent variables. Show that your differential is...
-
From our discussions of recent criticisms of research into cultural differences: Can we safely assume the existence of single national cultures? Is the influence of culture as an explanatory...
-
How can our trade theory of previous chapters be extended to incorporate changes in the nation's factor endowments, technology, and tastes? Is the resulting trade theory a dynamic theory of...
-
a. Develop a linear trend equation for the following data on demand for white bread loaves at a bakery (use of Excels Trendline, with display Equation on chart option, is recommended), and use it to...
-
True or False. The element mass matrices are always singular.
-
Upon hearing that Ross White (see Problems 6-25 and 6-26) is considering producing the brackets in house, the vendor has notified Ross that the purchase price would drop from $15 per bracket to...
-
Pharmacy sends a bag with heparin 25,000 units/250 mL NS. What rate (mL/hr) would the nurse set the infusion pump for?
-
1. How comparable are the two different methods? In what ways are they similar? In what ways are they different? 2. What are the positive and negative aspects of each approach that Shocker should...
-
A 500 W Carnot engine operates between constant temperature reservoirs at 100oC and 60.0oC. What is the rate at which energy is (a) Taken in by the engine as heat and (b) Exhausted by the engine as...
-
A Carnot engine is set up to produce a certain Work W per cycle. In each cycle, energy in the form of heat QH is transferred to the working substance of the engine from the higher-temperature thermal...
-
For 2023, Sevilla Corporations AFSI exceeds its regular taxable income, yet it owes no AMT. Can you explain this result?
-
Analyse the scenario and discuss whether a blockchain solution would be appropriate for this domain. You must refer to one or more of the Blockchain Suitability flowcharts You cant just simply answer...
-
How much tax will Hanna pay on her qualified dividends and capital gains?
-
The finance controller is seeking process improvement to reduce customer complaints due to late billings. The concerned departments include customer service who receives and processes orders,...
-
January Taxes February Taxes March Taxes Q 1 Earnings FWT $ 1 2 , 4 0 0 $ 1 2 , 7 2 0 $ 1 2 , 1 2 0 $ 3 3 1 , 2 0 0 Social Security $ 7 , 5 7 9 . 5 0 $ 7 , 2 4 0 . 3 6 $ 7 , 9 9 0 . 1 3 $ 3 6 7 , 9 0...
-
1. A note made on January 4 and due in 90 days would mature on what date? (assume February has 28 days) a. April 2 c. April 4 b. April 3 d. April 5 2. The maturity value of a $5,000, 60-day, 6...
-
A particle carrying \(+3.00 \mathrm{nC}\) of positive charge is at the origin of an \(x y\) coordinate system. Take \(V(\infty)\) to be zero for these calculations. (a) Calculate the potential \(V\)...
-
You've been asked to take over leadership of a group of paralegals that once had a reputation for being a tight-knit, supportive team, but you quickly figure out that this team is in danger of...
-
During a visit to the International Space Station, an astronaut was positioned motionless in the center of the station, out of reach of any solid object on which he could exert a force. Suggest a...
-
You have several identical balloons. You experimentally determine that a balloon will break if its volume exceeds 0.900 L. The pressure of the gas inside the balloon equals air pressure (1.00 atm....
-
A Jaguar XK8 convertible has an eight-cylinder engine. At the beginning of its compression stroke, one of the cylinders contains 499 cm3 of air at atmospheric pressure (1.01 X 105 Pa) and a...
-
A welder using a tank of volume 0.0750 m' fills it with oxygen (molar mass 32.0 g/mol) at a gauge pressure of 3.00 X 105 Pa and temperature of 37.0C. The tank has a small leak, and in time some of...
-
Write a program to create a file named "MyFile.txt". Store the following information in the file. Name Mlido Zodwa Phumla Tom Surname Samsons Bardwell Harrison Mpisane ITS Number 402106372 402103053...
-
of estion 2.1 Write a C++ program that calculates a customer's bill for a local cable company. The cable company has two types of customers: residential and business with different rates allocated...
-
Use Sobel mask and find horizontal and vertical gradient outputs for the given 5x5 image matrix which is coded 4-bit/pixel. (Don't use padding operation, obtain your output matrix as 3x3 image size!)...

Study smarter with the SolutionInn App


