Find G(jw) for the magnitude characteristic shown infigure. IGI -20 dB/dec 20 dB (%3 0.1 -20 dB/dec
Question:
Find G(jw) for the magnitude characteristic shown infigure.
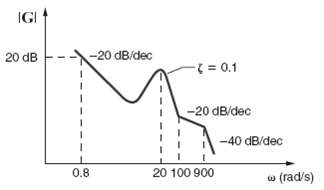
Transcribed Image Text:
IGI -20 dB/dec 20 dB (%3 0.1 -20 dB/dec -40 dB/dec 0.8 20 100 900 w (rad/s)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 87% (16 reviews)
Poles exist at 900 rad s zeroes exist at double zero at ...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Electrical Engineering questions
-
Find H (jw) if its magnitude characteristic is shown in fig 27.
-
Find H (jw) if its magnitude characteristic is shown in fig 28.
-
Find H(jw) if its magnitude characteristic is shown infigure. IHI +20 dB/dec -40 dB/dec 40 dB -20 dB/dec 10 80 120 w (rad/s)
-
9. Write a program to solve function given below, with the x and y value proposed by the user. And, based on the result, show "imaginary result" response for the case of the result of the function is...
-
A person floats in a boat in a small backyard swimming pool. Inside the boat with the person are some bricks. (a) If the person drops the bricks overboard to the bottom of the pool, does the water...
-
What techniques can help make the Internet search process more efficient?
-
Nancy Carpenter Optical Dispensary purchased supplies on account for \($1,200\). Two weeks later, the business paid half on account. Requirements 1. Journalize the two transactions for Nancy...
-
Carl and Carol have salaries of $14,000 and $22,000, respectively. Their itemized deductions total $8,500. They are married and both are under age 65. a. Compute their taxable income assuming they...
-
Question 8 of 12 < D Cullumber Company Balance Sheets December 31 2025 2024 Cash $19,500 $39,000 Accounts receivable (net) 75,000 54,000 Inventory 61,000 48,000 Plant assets (net) 240,000 230,000...
-
Which statement is true when customers pay bills with a credit card? Question 1 options: a) You can use QuickBooks' Merchant Account Service to get online authorization and then download payments...
-
Determine H(jw) if its magnitude characteristic is shown in figure -40 dB/dec IHI -20 dB/dec O dB -40 dB/dec 5 10 50 w (rad/s)
-
The series RLC circuit in figure is driven by a variable-frequency source. If the resonant frequency of the network is selected as w0 = 1600 rad/s, find the value of C. In addition, compute the...
-
Shannon and Jasmine bowl as a team. Each of them bowls three games in a tournament. They would like to know their individual averages for their three games and the team average. Allow the user to...
-
Plaintiff David Krasner brings this action against his former employer, HSH Nordbank AG (HSH), and his supervisor while employed there, Roland Kiser, alleging [among other things] sexual...
-
On December 16, 2005, James M. Eaton, Jr., and Marguerite Eaton [his mother] * * * fi led a complaint against [Bobby Joe] Waldrop alleging, among other things, that Waldrop had fraudulently induced...
-
Iberia Surgical [LLC] was formed, in August 1998, by a group of physicians practicing in Iberia Parish [Louisiana] for the purpose of establishing an ambulatory, outpatient surgery center. * * * Dr....
-
Mohammad Salim bought * * * a convenience store and gas station * * * . He made some improvements to the property and then offered it for sale. Talat Solaiman and Sabina Chowdhury approached Salim...
-
Rick Saldona began working as a traveling salesperson for Aimer Winery in 1988. Sales constituted 90 percent of Saldonas work time. Saldona worked an average of fi fty hours per week but received no...
-
At Enron Energy Services, many of the contracts were overvalued, as the energy loads required by its customers were based on guesswork and excessively optimistic assumptions. True/False
-
What are three disadvantages of using the direct write-off method?
-
Brown reports on a British Safety Council study that warns that employers good intentions could lead to ineffective employee well-being initiatives. Despite their enthusiasm, employers are often...
-
A 1-m3 tank is filled with a gas at room temperature 20C and pressure 100 kPa. How much mass is there if the gas is a) air, b) neon or c) propane?
-
A rigid tank of 1 m3 contains nitrogen gas at 600 kPa, 400 K. By mistake someone lets 0.5 kg flow out. If the final temperature is 375 K what is then the final pressure?
-
A cylindrical gas tank 1 m long, inside diameter of 20 cm, is evacuated and then filled with carbon dioxide gas at 25C. To what pressure should it be charged if there should be 1.2 kg of carbon...
-
The normal freezing point of ethanol (CH3CH2OH) is -117.30 C and its Kfp value is 1.99 C/m. A nonvolatile, nonelectrolyte that dissolves in ethanol is testosterone . How many grams of testosterone,...
-
Farmhill Ltd . had 1 , 5 6 5 , 3 0 0 common shares outstanding on 1 January 2 0 X 6 , the beginning of its 2 0 X 6 fiscal year. During the year, on 1 May, the company issued 5 0 9 , 0 0 0 preferred...
-
Be sure to answer all parts. Balance the following equations and write the corresponding ionic and net ionic equations (if appropriate). Include phase abbreviations: Part 1 out of 2 (a) Provide the...

Study smarter with the SolutionInn App


