The series RLC circuit in figure is driven by a variable-frequency source. If the resonant frequency of
Question:
The series RLC circuit in figure is driven by a variable-frequency source. If the resonant frequency of the network is selected as w0 = 1600 rad/s, find the value of C. In addition, compute the current at resonance and at w0/4 and4w0.
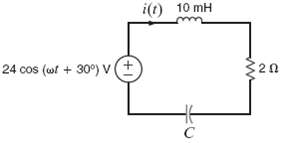
Transcribed Image Text:
i(t) 10 mH 24 cos (wt + 30°) v (+
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 63% (11 reviews)
Wo 1600 rad s 600 C JLC ...View the full answer

Answered By

Branice Buyengo Ajevi
I have been teaching for the last 5 years which has strengthened my interaction with students of different level.
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Related Video
In a series circuit, the same amount of current flows through all the components placed in it. On the other hand, in parallel circuits, the components are placed in parallel with each other and the circuit splits the current flow.
Students also viewed these Electrical Engineering questions
-
A parallel RLC circuit, which is driven by a variable frequency 2-A current source, has the following values: R = 1k, L = 100mH, and C = 10F. Find the bandwidth of the network, the half-power...
-
A parallel RLC circuit, which is driven by a variable frequency 2-a current source, has the following values: R=1Kohm,L=100mH, and C=10F. Find the bandwidth of the network, the half-power...
-
The shaft shown in the figure is driven by a gear at the right keyway, drives a fan at the left keyway, and is supported by two deep-groove ball bearings. The shaft is made from AISI 1020 cold-drawn...
-
If the molecular weight of air is 28.9, what is the density of air at atmospheric pressure and a temperature of 328.2 K? 1 atm = 1.013 x 10 5 N/m, the mass of a proton is 1.67262 x 10 -27 kg,...
-
A weather glass, as shown in FIGURE 15-45, is used to give an indication of a change in the weather. Does the water level in the neck of the weather glass move up or move down when a low-pressure...
-
How has the process of research changed in recent years? How have the changes been both beneficial and detrimental?
-
Lakeview Sales Consultants completed the following transactions during the latter part of March: Requirement 1. Journalize the transactions of Lakeview Sales Consultants. Include an explanation with...
-
A companys simplified balance sheet and income statement follow. Total assets and owners equity at the beginning of 2014 were $180,000 and $140,000, respectively. The owner made no investments or...
-
1. Write down an additional 1 negative effect of increased business competition in the middle of the COVID 19 pandemic due to crowded e-commerce space in a business. This should not be the same in...
-
From the adjustments columns in Exercise 5-9A, journalize the four adjusting entries, as of December 31, in proper general journal format. Exercise 5-9A Jim Jacobs Furniture Repair Work Sheet...
-
Find G(jw) for the magnitude characteristic shown infigure. IGI -20 dB/dec 20 dB (%3 0.1 -20 dB/dec -40 dB/dec 0.8 20 100 900 w (rad/s)
-
A series RLC circuit resonates at 1000 rad/s. If C = 20F, and it is known that the impedance at resonance is 2.4, compute the value of L, the Q of the circuit, and the band width.
-
How can we differentiate between the hypothesis that education increases productivity and the hypothesis that education is a signal for the workers innate ability?
-
Plaintiff is a company that owns a shopping plaza called Lammert Center located at 88018845 Ladue Road, St. Louis, Missouri. Defendant leased a portion of Lammert Center and operated a grocery...
-
In 1994, [Alvin Gebhart] began working at Mutual of New York (MONY) in San Diego, where he sold annuities and mutual funds. While at MONY, Gebhart met Jack Archer, a fellow MONY salesperson. In 1995,...
-
Petitioner, Leegin Creative Leather Products, Inc. (Leegin), designs, manufactures, and distributes leather goods and accessories. In 1991, Leegin began to sell [products] under the brand name...
-
Read the excerpt and answer the following questions. (a) Issue: On what issue did the Court focus? (b) Rule of Law: What does the Fifth Amendment to the U.S. Constitution, which the Court applied,...
-
Robert Wagner and Natalie Wood (the Wagners) entered into an agreement with Spelling-Goldberg Productions (SGP) relating to Charlies Angels (herein called the series). The contract entitled the...
-
When a company guarantees the debt of its unconsolidated affiliates, it is a signal that these entities may be used to raise debt without recording it on the sponsoring companys balance sheet....
-
You are thinking of investing in one of two companies. In one annual report, the auditors opinion states that the financial statements were prepared in accordance with generally accepted accounting...
-
How feasible do you think it is for SMEs to use HR analytics? What problems might they face?
-
A glass is cleaned in 45oC hot water and placed on the table bottom up. The room air at 20oC that was trapped in the glass gets heated up to 40oC and some of it leaks out so the net resulting...
-
A hollow metal sphere of 150-mm inside diameter is weighed on a precision beam balance when evacuated and again after being filled to 875 kPa with an unknown gas. The difference in mass is 0.0025 kg,...
-
A vacuum pump is used to evacuate a chamber where some specimens are dried at 50C. The pump rate of volume displacement is 0.5 m3/s with an inlet pressure of 0.1 kPa and temperature 50C. How much...
-
Compare Income Statements and Balance Sheets of Competitors a. Following are selected income statements from two pharmaceutical companies, Pfizer and Dr. Reddy's, for their respective 2018 fiscal...
-
Describe how the nuclear process compares to a similar chemical process in terms of energy ? what is the anatomical pathway and physical and chemical processes of a cookie. starting at the mouth and...
-
Sodium hydroxide is used extensively in acid-base titrationsbecause it is a strong, inexpensive base. A sodium hydroxidesolution was standardized by titrating 26.62 mL of 0.1007 Mstandard...

Study smarter with the SolutionInn App


