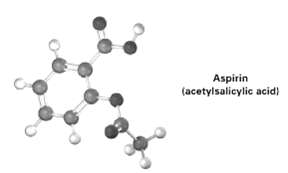
Following is a molecule model of aspirin (acetylsalicylic acid). Identify the hydribization of each carbon atom in
Question:
Following is a molecule model of aspirin (acetylsalicylic acid). Identify the hydribization of each carbon atom in aspirin, and tell which atoms have lone pairs of electrons (gray = c. red = O, ivory =H).
Transcribed Image Text:
Aspirin (acetylsalicylic acid)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (10 reviews)
010 IU CIH v0 O O 6 sp Aspirin ...View the full answer

Answered By

Larlyu mosoti
I am a professional writer willing to do several tasks free from plagiarism, grammatical errors and submit them in time. I love to do academic writing and client satisfaction is my priority. I am skilled in writing formats APA, MLA, Chicago, and Harvard I am a statistics scientist and I can help out in analyzing your data. I am okay with SPSS, EVIEWS, MS excel, and STATA data analyzing tools.
Statistical techniques: I can do linear regression, time series analysis, logistic regression, and some basic statistical calculations like probability distributions. . I'm ready for your working projects!
Services I would offer:
• Academic writing.
• Article writing.
• Data entry.
• PDF conversion.
• Word conversion
• Proofreading.
• Rewriting.
• Data analyzing.
The best reason to hire me:
- Professional and Unique work in writing.
- 100% satisfaction Guaranteed
- within required time Express delivery
- My work is plagiarism Free
- Great communication
My passion is to write vibrantly with dedication. I am loyal and confident to give my support to every client. Because Client satisfaction is much more important to me than the payment amount. A healthy client-contractor relationship benefits in the longer term. Simply inbox me if you want clean work.
5.00+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
What is the hydribization of each carbon atom in acetonitrile (Problem 1.25)?
-
Tell the number of hydrogens bonded to each carbon atom in the following substances and give the molecular formula ofeach: OH H (a) (b) CO2CH3 Ephedrine Cocaine
-
Describe the hybrid orbitals used by each carbon atom in the following molecules: a. b. C-C-C-OH
-
Like Death Valley, the Dead Sea is a place where you can walk on dry land below sea level, and an apparently unmotivated hole in the ground along a strike-slip fault. How do you think it formed?
-
Determine the percentage of vesting for the following employees. See Exhibits 19.1 and 19.2. a. Glenda has five years of service completed as of September 23, 2016, her employment anniversary date....
-
A system has four processes and five allocatable resources. The current allocation and maximum needs are as follows: What is the smallest value of x for which this is a safe state? Process A Process...
-
How large (MJ/h) should a heat-rejection system be if it must dump the entire heat production of a \(1000 \mathrm{~m}^{2}\) solar collector array in Denver, CO, on August 21 if the collector is at...
-
Forty-five kilograms of a solution containing 30 wt% ethylene glycol in water is to be extracted with furfural. Using Figures 4.14a and 4.14c, calculate:(a) The minimum quantity of solvent.(b) The...
-
Two agents meet to trade two goods. Agent 1 has preferences representable by utility func- tion u (x()) = x() (x()), while agent 2's preferences are representable by utility function u (x(2)) (x(2))...
-
During the financial year ended 30 June 2004, M Bhd (MB) which has a 40% ownership interest in its associate N Bhd (NB) purchased merchandize from the associate amounting to RM1,500, 000. At the end...
-
Draw a line-bond structure for 1, 3-butadiene, H2C = CH CH = CH2; indicate the hydribization of each carbon; and predict the value of each bond angle.
-
Draw a line-bond structure for the propyne, CH3C CH; indicate the hydribization of each carbon; and predict a value for each bond angle.
-
The market price for Potash Corp. of Saskatchewan Inc. closed at $114.39 and $154.45 on December 31, 2009, and December 31, 2010, respectively. The dividends per share were $0.15 for 2009 and $0.14...
-
1. What is the ratio of 2 lengths which are 25 cm and 7.5 m respectively?
-
Imagine yourself at a fair playing one of the midway games. Pick a game and calculate the expected value and post your results along with how you calculated them.
-
(a). Company Inc., private consumer regulator firm, claims that the standard deviation of the rating of Formula One races in the town main car racing circuit was at least 15.34. (b). An outside...
-
Determine how many tablets will be needed to give the dosage. Prepare a dosage of 6.4 mg using tablets with a strength of 1.6 mg.
-
What choice is a multiple of 5? a) 93 b) 63 c) 65 d) 84
-
All 100 first-year students at a small college take three courses in the Core Studies program. Two courses are taught in large lectures, with all 100 students in a single class. The third course is...
-
Solve each equation. x 3 - 6x 2 = -8x
-
a. In vapor-liquid equilibrium, mixtures sometimes occur in which the compositions of the coexisting vapor and liquid phases are the same. Such mixtures are called azeotropes. Show that a binary...
-
On the basis of the rule that anything unusual about a structure must be shown explicitly, the nitrogen in the structure NH 3 is seen to have an unshared pair of electrons whether these electrons are...
-
Ozone O 3 is a form of oxygen found in the upper atmosphere. It has the connectivity O OO and is neutral. (a) Show a Lewis structure for ozone. (b) Calculate the formal charge on each oxygen of...
-
(a) Draw a Lewis structure for the carbonate anion CO 3 2 each oxygen is bonded only to the carbon. (b) Calculate the formal charge on each atom. (c) What is the shape of this species? (d)...
-
A positive charge q = +8 nC is at the origin, and a second positive charge q = +12 nC is on the x axis at a = 4 m, Find the net electric field (a) at point P, on the x axis at x=7 m, and (b) at point...
-
8. Hooten Carpentry had the following accounts and account balances after adjusting entries. Assume all accounts have normal balances. Prepare the adjusted trial balance for Hooten Carpentry as of...
-
Explain at least one major difference that exists today between US GAAP and IFRS on the accounting for foreign currency transactions. Be specific.

Study smarter with the SolutionInn App


