From Figure, find (a) The temperature at which water boils on a mountain where the atmospheric pressure
Question:
From Figure, find
(a) The temperature at which water boils on a mountain where the atmospheric pressure is 70kPa,
(b) The temperature at which water will boil in a container in which the pressure has been reduced to 0.5 atm, and
(c) The pressure at which water will boil at115oC.
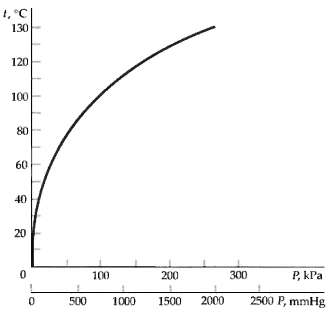
Transcribed Image Text:
1, °C 130 120 100 80 60 40 20 P, kPa 100 200 300 2500 P, mmHg 2000 500 1000 1500
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (9 reviews)
a At 70 kPa the boilin...View the full answer

Answered By

Danish Sohail
My objective is to become most reliable expert for clients. For last 10 years I have been associated with the field of accounting and finance. My aim is to strive for best results and pay particular attention to client needs. I am always enthusiastic to help clients for issues and concerns related to business studies. I can work on analysis of the financial statements, calculate different ratios and analysis of ratios. I can critically evaluate stock prices based on the financial analysis and valuation for companies using financial statements of the business entity being valued with use of excel tools. I have expertise to provide effective and reliable help for projects in corporate finance, equity investments, financial accounting, cost accounting, financial planning, business plans, marketing plans, performance measurement, budgeting, economic research, risk assessment, risk management, derivatives, fixed income investments, taxation, auditing, and financial performance analysis.
4.80+
78+ Reviews
112+ Question Solved
Related Book For 

Fundamentals of Ethics for Scientists and Engineers
ISBN: 978-0195134889
1st Edition
Authors: Edmund G. Seebauer, Robert L. Barry
Question Posted:
Students also viewed these Thermodynamics questions
-
The temperature at which water boils (the boiling point) depends on elevation: The higher the elevation, the lower the boiling point will be. At sea level, water boils at 212F; at an elevation of...
-
The temperature at which water starts to boil is also linearly related to barometric pressure. Water boils at 212F at a pressure of 29.9 in Hg (inches of mercury) and at 191F at a pressure of 28.4 in...
-
A manufacturer of candy must monitor the temperature at which the candies are baked. Too much variation will cause inconsistency in the taste of the candy. Past records show that the standard...
-
Consider the pistoncylinder arrangement shown in the sketch below. The gas forces on each side of the piston, assuming there are no frictional forces at the pistoncylinder interface, balance the...
-
Steve Drake sells a rental house on January 1, 2016, and receives $120,000 cash and a note for $45,000 at 10 percent interest. The purchaser also assumes the mortgage on the property of $35,000....
-
The following were selected from among the transactions completed by Babcock Company during November of the current year: Nov. 3 Purchased merchandise on account from Moonlight Co., list price...
-
If a company loses a potential major order, what should sales management do to alleviate the risk of this happening again?
-
The Hardware Warehouse is evaluating the safety stock policy for all its items, as identified by the SKU code. For SKU M4389, the company always orders 80 units each time an order is placed. The...
-
What is the relationship between values, power, and politics in nonprofit organizations? Question 2: How might the level of reliability and the level of autonomy for funding sources affect the...
-
6. Bill and Caroline's budget for a typical month is shown. CC Bill INCOME (S) Caroline Total EXPENSES (S) Fixed Expenses Mortgage Telephone Cable Internet Loan payment Car payment Car insurance...
-
(a) Calculate the volume of 1 mol of steam at 100 o C and a pressure of 1 atm, assuming that it is an ideal gas. (b) Find the temperature at which the steam will occupy the volume found in part (a)...
-
The van der Waals constants for helium are a = 0.03412 L2atm/mol2 and b = 0.0237 L/mol. Use these data to find the volume in cubic centimeters occupied by one helium atom and to estimate the radius...
-
Let the pdf f(x; 1 , 2 ) be of the form exp[p 1 ( 1 , 2 )K 1 (x) + p 2 ( 1 , 2 )K 2 (x) + H(x) + q 1 ( 1 , 2 )], a < x < b, zero elsewhere. Suppose that K' 1 (x) = cK' 2 (x). Show that f(x; 1 ,...
-
Explain how the central banks balance sheet changes.
-
Analyze the link between the monetary base and the money supply.
-
Dollarizing and joining a monetary union both involve giving up a countrys own currency, but there are key differences between these two options. Identify one factor that might lead a country to...
-
The collapse of banks and the banking system disrupts both the payments system and the screening and monitoring of borrowers. a. Intermediaries are insolvent when their liabilities exceed their...
-
Describe unconventional monetary policy tools and how they work.
-
A switch from straight-line to accelerated depreciation would: A. increase the NPV and decrease the first year operating income after taxes. B. increase the first year operating income after taxes...
-
How has the too-big-to-fail policy been limited in the FDICIA legislation? How might limiting the too-big-to-fail policy help reduce the risk of a future banking crisis?
-
When a pure substance melts, it (a) Undergoes a chemical change. (b) Changes state. (c) Changes from a liquid to a solid. (d) Sublimes.
-
The following measurements were made on a Thyrite resistor: (a) Graph V ab as a function of I. (b) Does Thyrite obey Ohm's law? How can you tell? (c) Graph the resistance R = Vab/I as a function of...
-
The following measurements of current and potential difference were made on a resistor constructed of Nichrome wire: (a) Graph Vab as a function of I. (b) Does Nichrome obey Ohm's law? How can you...
-
A resistor with a 15.0-V potential difference across its ends develops thermal energy at a rate of 327 W. (a) What is its resistance? (b) What is the current in the resistor?
-
Rewrite the Roman numeral using the least number of symbols possible and the subtractive principle where applicable?
-
Determine, with justification, the smallest possible average of a set whose elements are distinct positive integers that includes the integer 200.
-
This study is for students joining University who are not living in the region. Suppose that a student will join the University next semester and wants to choose between renting a one-bedroom...

Study smarter with the SolutionInn App


