From what epoxide and what nucleophile colld each of the following compound be prepared Inppued? (Assums each
Question:
From what epoxide and what nucleophile colld each of the following compound be prepared Inppued? (Assums each is racemic.)
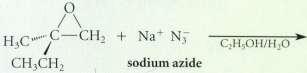
Transcribed Image Text:
C,H OH/H,O CH CH2 sodium azide
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (15 reviews)
The strategy in this problem is to let the OH group originate fro...View the full answer

Answered By

Ehsan Mahmood
I’ve earned Masters Degree in Business Studies and specialized in Accounts & Finance. Couple with this, I have earned BS Sociology from renowned institute of Pakistan. Moreover, I have humongous teaching experience at Graduate and Post-graduate level to Business and humanities students along with more than 7 years of teaching experience to my foreign students Online. I’m also professional writer and write for numerous academic journals pertaining to educational institutes periodically.
4.90+
248+ Reviews
287+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Each of the following compounds has a nitrogennitrogen bond: N2, N2H4, N2F2. Match each compound with one of the following bond lengths: 110 pm, 122 pm, 145 pm. Describe the geometry about one of the...
-
Each of the following compounds is an aromatic compound bearing a substituent that we did not discuss in this chapter. Using the principles that we discussed in this chapter, predict the major...
-
Can the following compound be prepared via a Williamson ether synthesis? Explain your answer.
-
The portfolio of stock that comprises the ASX200 index is currently worth $5000. The continuously compounded interest rates on Australian government bonds is 1.5% per annum for each of the next five...
-
Assume initially that Demand and supply for premium coffees (one pound-bags) are in equilibrium. Now assume Starbucks introduces the world to premium blends so Demand rises substantially. Describes...
-
An aluminum calorimeter with a mass of 100 g contains 250 g of water. The calorimeter and water are in thermal equilibrium at 10.0C. Two metallic blocks are placed into the water. One is a 50.0-g...
-
An experimenter wants to compare the time to failure \(y\) after rebuilding a robotic welder by three different methods but adjusting for the covariate \(x=\) age of robotic welder. Suppose the data,...
-
Ithaca Snowboards Corporation is a large U.S. producer of in-style winter recreational equipment and apparel. Ithaca Snowboards currently has its primary facilities in the United States as well as...
-
post a 150-300-word response to the following question. Also add a reference with your response (e.g. course textbook). Abnormal Psychology Why is it important to understand the history of abnormal...
-
You plan to open a small business for manufacturing pet collars, leashes, and harnesses. You have found a workshop space you can use for sewing your products. After some research and planning, you...
-
What products are formed when gach of the following ethers reacts with concenffated aqueous HI? 2-ethoxy-2,3-dimethylbutane
-
Suggest a Williamron other cynthosis, if one is possible, for each of the following compounds. If no Williamson ether synthesis is possible, explain why. (CH3)2CH---S---CH3
-
Consider the following reaction: It takes 2.00 L of pure oxygen gas at STP to react completely with a certain sample of aluminum. What is the mass of aluminum reacted? 4Al(s) + 30(g) - 2AlO3(s)
-
A (a cash-basis individual) forms JUF Corporation by transferring $50,000 cash in exchange for $50,000 of stock. B (a cash-basis individual) transfers two assets: (1) a capital asset with a basis of...
-
On April 7, 2023, an auditor noticed that UGL Inc. accidentally recorded a rent expenditure of $41,000 as an expense instead of as prepaid. The purchase was made on the last day of the 2022 fiscal...
-
Nortex Chemicals, Inc. is involved in the natural gas processing industry in the Texas Panhandle. One of its primary operations involves purchasing large tanks, each roughly the size of a rail car,...
-
1. An optometrist has very limited space in her office. The office is 5.0 m in length, but the eye chart has directions that state that the patient must be 8 m away from the chart. How does the...
-
A-7 HILBERT TRANSFORM PAIRS+ Definition of Hilbert Transform: x(t) = x(t) * Function 1. x(at + b) 2. x(t) + y(t) dnx(t) 3. dtn 4. A constant 5. 1 t 6. sin (wot + 0) 7. sin at at 8. ejwot 9. 8 (t) =...
-
What advice does the person you are interviewing have for newcomers entering this field? What can you do now to get yourself started on a career path toward this position?
-
Danielle has an insurance policy with a premium of $75 per month. In September she is in an accident and receives a bill worth $2990 for the repair of her own property. Her deductible is $250 and her...
-
Provide a reaction mechanism for step 1, formation of the imine, in Eq. 23.22. EtNH + H3C-C-CH3 acetone -HO [HC- NEt H3C-C-CH3 an imine (not isolated) H, Pt 30 psi EtOH NHET H3C-CH-CH3 (23.22)...
-
Provide reaction mechanisms for the following equations. (a) Eq. 23.17 (b) Eq. 23.18 (c) Suggest a reason why the reaction in Eq. 23.18 stops after two additions, and a third doesnt occur in high...
-
Using their solubilities in acidic or basic solution, design a separation of p-chlorobenzoic acid, p-chloroaniline, and p-chlorotoluene from a mixture containing all three compounds.
-
So I'm new to arduino, and I am working on a project for school and I was wondering if I am correct in my wiring and coding. The premise of the project is an automatic pet feeder. Basically, a pet...
-
Image transcription text Wind tunnel measurements of the pressure and skin friction around a NACA 2415 airfoil at 8 degrees angle of attack resulted in the following data of pressure and skin...
-
Image transcription text The following table contains load-extension data from a tensile test on a cylindrical specimen with gauge length 9mm and gauge diameter 5mm. Load-extension Data Load [KN] 0...

Study smarter with the SolutionInn App


