Provide a reaction mechanism for step 1, formation of the imine, in Eq. 23.22. EtNH + H3C-C-CH3
Question:
Provide a reaction mechanism for step 1, formation of the imine, in Eq. 23.22.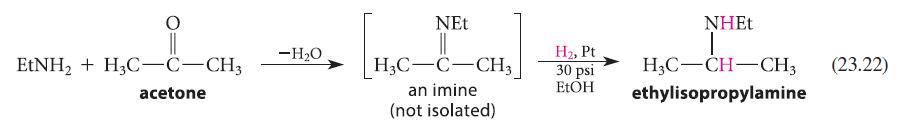
Transcribed Image Text:
EtNH₂ + H3C-C-CH3 acetone -H₂O [H₂C- NEt H3C-C-CH3 an imine (not isolated) H₂, Pt 30 psi EtOH NHET H3C-CH-CH3 (23.22) ethylisopropylamine
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (4 reviews)
The reaction of a ...View the full answer

Answered By

Emily Grace
With over a decade of experience providing top-notch study assistance to students globally, I am dedicated to ensuring their academic success. My passion is to deliver original, high-quality assignments with fast turnaround times, always striving to exceed their expectations.
4.90+
3+ Reviews
22+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
(a) A synthesis of pyrimidine is outlined below (i) Give a mechanism for Step 1. (5 marks) (ii) Give a mechanism for Step 2. (2 marks) (b) Give the structure of the product formed in the following...
-
The reaction of ninhydrin with an ?-amino acid occurs in several steps. (a) The first step is formation of an imine by reaction of the amino acid with ninhydrin. Show its structure and the mechanism...
-
Simple sugars undergo reaction with phenyl hydrazine, PhNHNH2, to yield crystalline derivatives called osazones. The reaction is a hit complex however, as shown by the fact that glucose and fructose...
-
What are the values of the engineering economy symbols P, F, A, i, and n in the following functions? Use a question mark for the symbol that is to be determined. (a) NPER (8%, 1500,8000,2000) (b) FV...
-
Identify the six major steps of the accounting process and explain each step.
-
For what reasons do businesses withhold funds from their employees pay?
-
A university pumps its water from wells located on campus. The falling water table has caused pumping costs to increase, the quantity of water available to decrease, and the quality of water to...
-
Boyne Inc. had beginning inventory of $12,000 at cost and $20,000 at retail. Net purchases were $120,000 at cost and $170,000 at retail. Net markups were $10,000; net markdowns were $7,000; and sales...
-
as Suppose X is a normal variable with mean and variance 2. Let f(x) be the density function of X. For a constant 0, define the tilted density function fo(x) = f(x)ex Mo where Me is the normalization...
-
Why is base (NaOH or KOH) added as a second step in each of the reactions shown in Eqs. 23.23 and 23.24? PhCH+H_NC(CH3)3 benzaldehyde tert-butylamine + Me,NH dimethylamine cyclohexanone NaBH(OAC) 3...
-
Provide reaction mechanisms for the following equations. (a) Eq. 23.17 (b) Eq. 23.18 (c) Suggest a reason why the reaction in Eq. 23.18 stops after two additions, and a third doesnt occur in high...
-
Is P2 spanned by 1 + x, x + x2, 1 + x2?
-
While analyzing the economics of health care, consider the many factors in forming a health policy. Answer the following questions: Make a recommendation - how should health care be financed? What do...
-
When octane (C8H18, density = 0.699 g/mL) undergoes complete combustion, it provides energy according to the reaction: 2 C8H18 (1) + 25 O2(g) 16 CO(g) + 18 HO(1) = AH -10,392 kJ How much heat, in...
-
A family member is thinking about funding his granddaughters university education in 8 years when she is expected to enroll at UWI, St. Augustine. He opens a special savings account, where he can...
-
Sample of short script of a two persons who are imagining and acting out a scene Consider a scene involving two characters in action (i.e., negotiating, fighting/ quarreling. debating, convincing,...
-
There was a prosecuting attorney who had a person on trial for an infraction of the law for which there was a lot of circumstantial evidence. The prosecuting attorney is reason- ably sure that he...
-
Describe the principal steps in the analysis phase. What are the major deliverables?
-
In Problems 718, write the augmented matrix of the given system of equations. f0.01x0.03y = 0.06 [0.13x + 0.10y = 0.20
-
Predict the products of the following acid-base reactions. If the equilibrium would not result in the formation of appreciable amounts of products, you should so indicate. In each case label the...
-
Show how you might synthesize ethynylbenzene from methyl phenyl ketone.
-
Outline all steps in a synthesis of propyne from each of the following: (a) CH3COCH3 (b) CH3CH2CHBr2 (c) CH3CHBrCH2Br (d) CH3CH==CH2
-
The information presented here represents selected data from the December 31, 2022, balance sheets and income statements for the year then ended for three firms. Required: Calculate the missing...
-
In the context of lysosomal storage disorders, what specific molecular defects lead to the aberrant accumulation of macromolecules within lysosomes, and how do these conditions manifest clinically?
-
Carly Rae Jepson realized that she had roughly 15 minutes, so she decided to invest some of the earnings from her (surprisingly) hit single, Call Me Maybe in fixed income securities. You gave her the...

Study smarter with the SolutionInn App


