Give IUPAC names for the followingcompounds: (b) HCHHCHCHCH3 (a) CCHH CH (e) CCHHH3 (d) CH2CH2CH3 CCH3CCH3C2
Question:
Give IUPAC names for the followingcompounds:
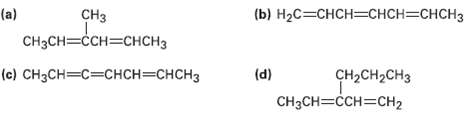
Transcribed Image Text:
(b) НаС—СHCH—СHCH—CHCH3 (a) Cнзсн—СCH—снсHз CHз (e) CнзCH—с—снсH—снсH3 (d) CH2CH2CH3 CнзCH3CCH3Cн2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 90% (11 reviews)
All of these compounds can exhibit EZ ...View the full answer

Answered By

Akshay Singla
as a qualified engineering expert i am able to offer you my extensive knowledge with real solutions in regards to planning and practices in this field. i am able to assist you from the beginning of your projects, quizzes, exams, reports, etc. i provide detailed and accurate solutions.
i have solved many difficult problems and their results are extremely good and satisfactory.
i am an expert who can provide assistance in task of all topics from basic level to advance research level. i am working as a part time lecturer at university level in renowned institute. i usually design the coursework in my specified topics. i have an experience of more than 5 years in research.
i have been awarded with the state awards in doing research in the fields of science and technology.
recently i have built the prototype of a plane which is carefully made after analyzing all the laws and principles involved in flying and its function.
1. bachelor of technology in mechanical engineering from indian institute of technology (iit)
2. award of excellence in completing course in autocad, engineering drawing, report writing, etc
4.70+
48+ Reviews
56+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Give IUPAC names for the following alkenes: (a) (b) (c) (d) (e) (f) OH CI
-
Give IUPAC names for the following compounds. (a) (b) (c) (d) (e) (f) Ph CH3C C CH CH H3C CH3 - (CH3)3C C-CH (CH3)CH2CH3 CH CHC CC-OH CH,CH CH C-C CH
-
Give IUPAC names for the following compounds. (a) (b) (c) CH3 CH CH CH CH, CH, CH CH
-
Given a shaky economy and high heating costs, more and more households are struggling to pay utility bills (The Wall Street Journal, February, 14, 2008). Particularly hard hit are households with...
-
What ethical standards for human resource management do you think a company should require from all its operations worldwide? In what areas of HRM, if any, should ethical standards be relaxed to...
-
The beaker on the right contains 0.1 M acetic acid solution with methyl orange as an indicator. The beaker on the left contains a mixture of 0.1 M acetic acid and 0.1 M sodium acetate with methyl...
-
When using statistical sampling for attributes, an auditor should assess control risk at the maximum assuming: a. The sample rate of deviation plus the allowance for sampling risk is less than the...
-
The Furry Friends Foundation is a non-profit organization that finds homes for abandoned animals that are suitable for adoption. FFF began operations with a bequest from a wealthy gentleman who lived...
-
Georgia Lemoy was one of three children. She never married or had children of her own. She and her two siblings all lived in the western suburbs of Chicago throughout their lives. The extended family...
-
Throughout the course, you will be working on a Wealth Management Analyst Project For this project, imagine you are a new hire at a wealth management firm and tasked with determining the location of...
-
The following model is that of an allylic carbocation intermediate formed by Protonation of a conjugated diene with HBr. Show the structure of the diene and the structures of the final...
-
What product(s) would you expect to obtain from reaction of 1, 3-cyclo-hexadiene with each of the following? (a) 1 mol Br2 in CH2C12 (b) O3 followed by Zn (c) 1 mol HCI in ether (d) 1 mol DCl in...
-
Castle Consulting Agency began business in February. The transactions entered into by Castle during its first month of operations are as follows: a. Acquired articles of incorporation from the state...
-
The volume of any spherical balloon can be found by using the formula. V = r Write an equation for r in terms of V and .
-
Morrow Corporation had only one job in process during May Job X 3 2 Z and had no finished goods inventory on May 1 . Job X 3 2 Z was started in April and finished during May, Data concerning that job...
-
Waterway, Inc. applies overhead to production at a predetermined rate of 90% of direct labor cost. Job No. 250, the only job still in process at the end of August, has been charged with manufacturing...
-
XYZ Corp. operates under ideal conditions of certainty. It acquired its sole asset on January 1, 20x1. The asset will yield $600 cash at the end of each year from 20x1 to 20x3, inclusive, after which...
-
Donald ( a married taxpayer filing jointly ) owns a wide variety of commercial rental properties held in a single member LLC . Donald's LLC reports rental income of $ 1 , 5 0 0 , 0 0 0 . The LLC pays...
-
You are investigating a rural accident. The evidence at the scene suggests that a car was traveling west when struck by a van traveling south. The crushed vehicles stuck together, then came to rest...
-
The following data are supplied for the common stocks of Nikola Corporation, Tesla, Inc. and General Motors: Nikola Corp (NKLA) Tesla Inc. (TSLA) Close Price ($) Close Price ($) 67.53 30.00 40.81...
-
How does the solubility of a gas in a liquid depend on temperature? How does this temperature dependence affect the amount of oxygen available for fish and other aquatic animals?
-
What would be the ratio of products in the reaction of chlorine with ethylbenzene if chlorine randomly abstracted a methyl or methylene proton? What is the reactivity ratio for the benzylic hydrogens...
-
Predict the major products when the following compounds are irradiated by light and treated with (1) 1 equivalent of Br2 and (2) excess Br2. (a) Isopropylbenzene (b) (tetralin)
-
Propose a mechanism for the reaction of benzyl bromide with ethanol to give benzyl ethyl ether (shown above).
-
Suresh Company reports the following segment (department) income results for the year. Sales: Expenses Avoidable Unavoidable Total expenses Income (loss) Department M $ 66,000 Department N $ 38,000...
-
EZ Tees Corporation produces T-shirts. The company uses a standard costing system and has set the following standards for direct materials and direct labor (for one shirt): Fabric (1.5 yds. @ $2.80)...
-
Question : List the competitive advantages of Nordstrom: the things that make it different from competitors in positive ways. Nordstrom: A Shining Example of Service Excellence 'Use your best...

Study smarter with the SolutionInn App


