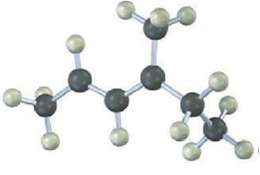
The following model is that of an allylic carbocation intermediate formed by Protonation of a conjugated diene
Question:
The following model is that of an allylic carbocation intermediate formed by Protonation of a conjugated diene with HBr. Show the structure of the diene and the structures of the final reactionproducts.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 61% (13 reviews)
HC H HC CH3 HC OH H Br C H CH3 or CH3 HC CHCH3 B...View the full answer

Answered By

GERALD KAMAU
non-plagiarism work, timely work and A++ work
4.40+
6+ Reviews
11+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The following model is that of an aldohexose: (a) Draw Fischer projections of the sugar, its enantiomer, and a diastereomer. (b) Is this a D sugar or an L sugar? Explain. (c) Draw the ? anomer of the...
-
The following model is specified:y 1 = 1 y 2 + 11 x 1 + 1 ,y 2 = 2 y 1 + 22 x 2 + 32 x 3 + 2 . All variables are measured as deviations from their means. The sample of 25 observations produces...
-
The following model is specified: y1 = 1y2 + 11x1 + 1, y2 = 2y1 + 22x2 + 32x3 + 2. All variables are measured as deviations from their means. The sample of 25 observations produces the following...
-
The following Excel output summarizes the results of an analysis of variance experiment in which the treatments were three different hybrid cars and the variable measured was the miles per gallon...
-
Suppose you work in the HR function at Nissan when it is identifying employees to work on the joint manufacturing project in Mexico. Briefly advise the company on how to prepare these employees to...
-
Two coils are mutually coupled, with L1 = 25 mH, L2 = 60 mH, and k = 0.5. Calculate the maximum possible equivalent inductance if: (a) The two coils are connected in series (b) The coils are...
-
The invention and commercialization of a new chemical is a significant undertaking. An example is the invention of nylon in the laboratory and its transformation to a nylon stocking. Please read the...
-
Relaxing Recliner Chairs completed the following selected transactions: 2013 Jul. 1 Sold merchandise inventory to Great Mart, receiving a $ 45,000, nine-month, 12% note. Ignore cost of goods sold....
-
Bartman, Corp. observes that the Swiss franc (SF) is quoted at $0.6164/SF, while the Swedish krona (SK) is quoted at $0.1981/SK. What is the SK/SF cross rate?
-
1. The following table shows the unit shipping cost between cities, the supply, demand and the destination city. Use the Minimum Cost Method to find the initial solution (or BFS) and then find the...
-
The following diene does not undergo Diels?Alder reactions. Explain.
-
Give IUPAC names for the followingcompounds: (b) HCHHCHCHCH3 (a) CCHH CH (e) CCHHH3 (d) CH2CH2CH3 CCH3CCH3C2
-
Revise the following to eliminate empty words. Except for the instance of MySpace, social networking sites are booming.
-
what is disruptive technology? Provide 2 example disruptive technologies with old technologies.
-
1 . ) What energy neutron has an associated wavelength of order the radius of a nucleus? 2 . ) Describe the position and momentum uncertainties for a harmonic matter - wave.
-
How group dynamics support or hinder team performance and explain the approach you will take to ensure that they support the team.
-
Comprehensive analysis of the impact that women in leadership roles might have on community and social change. Describe the leadership approaches and theories women in leadership roles might further...
-
How can managers determine if organizational structure is out of alignment with an organization's strategy in a criminal justice organization? If the organizational structure were misaligned, what...
-
Decide whether the FVT is applicable, and if so, find \(x_{s s}\). \(X(s)=\frac{2 s+\frac{1}{3}}{\left(s+\frac{1}{3} ight)\left(s^{2}+s+1 ight)}\)
-
Give the structural formulas of the alkenes that, on ozonolysis, give: a. (CH3)2C=O and CH2=O b. Only (CH3CH2)2C=O c. CH3CH=O and CH3CH2CH=O d. O=CHCH2CH2CH2CH=O
-
A solution is an equimolar mixture of two volatile components A and B. Pure A has a vapor pressure of 50 torr, and pure B has a vapor pressure of 100 torr. The vapor pressure of the mixture is 85...
-
Predict the major products of the following reactions. (a) toluene + excess Cl2 (heat, pressure) (b) benzamide (PhCONH2) + Na (;oqioed NH3, CH3CH2OH) (d) o-xylene + H2 (1000 psi, 100oC, Rh catalyst)...
-
Predict the major products of treating the following compounds with hot, concentrated potassium permanganate, followed by acidification with dilute HCl. (a) isopropylbenzene (b) p-xylene (c) tetralin)
-
Propose a mechanism for the bromination of ethylbenzene shown above.
-
Reflecting on the book Who Gets Promoted, Who Doesn't, and Why (Asher, 2008), answer the following questions in depth. Explain the statement, "all business is sales." What six things matter? If...
-
Reflecting on the book Who Gets Promoted, Who Doesn't, and Why (Asher, 2008), answer the following questions in depth. No matter what you have done in the past, the boss really doesn't care. Why?...
-
Do you think Richard Scrushy should have been found guilty or acquitted for his role in the case against HealthSouth? https://www.wsj.com/articles/SB111702610398942860

Study smarter with the SolutionInn App


