Give IUPAC names for the following compounds: (a) (b) (c) CH CHCHCH2CH CH2CH2CH3 H-CH2CHCH20H
Question:
Give IUPAC names for the following compounds:
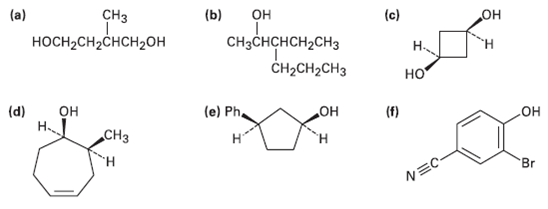
Transcribed Image Text:
(a) (b) (c) CHз он он CHзснCHCH2CHз CH2CH2CH3 носH-CH2CHCH20H но (d) Н (e) Ph. но° (f) Он СНз но н Br NEC-
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 64% (17 reviews)
a CH3 HOCHCHCHCHOH d 2Methyl14butanediol HOH CH3 ...View the full answer

Answered By

Divya Munir
I hold M.Sc and M.Phil degrees in mathematics from CCS University, India and also have a MS degree in information management from Asian institute of technology, Bangkok, Thailand. I have worked at a international school in Bangkok as a IT teacher. Presently, I am working from home as a online Math/Statistics tutor. I have more than 10 years of online tutoring experience. My students have always excelled in their studies.
4.90+
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Give IUPAC names for the following compounds: CH CH3CH2CH=CCH2CH3 (b) (a) CH CHCH-H CHCH2CH2CHCHCHCH2CH3 CH CH (d) (c) CHCHCHCHC CH
-
Give IUPAC names for the followingcompounds: H CHCH2CCH H CH CHH2CH2CH (b) (e) CHH2H2C CH (a) H-H H CH3CH2CHCH2CH,CHCH3 (e) CH H CHCH2CH2CHCH2CH CH CH-H>H3 (d)
-
Give IUPAC names for the following alkenes: (a) (b) (c) (d) (e) (f) OH CI
-
John dies on 3 March 2021. Between 6 April 2020 and 3 March 2021, he has capital gains of 1,200 and capital losses of 15,400. His net gains in recent tax years (and the annual exemption for each...
-
Rank each of the 15 grand strategy options discussed in this chapter on the following three scales: High --Low Cost High --Low Risk of failure High----- Potential for exceptional growth --Low
-
Let I := [0; /2] and let f : I R be defined by f(x) := sup{x2, cos x} for x I. Show there exists an absolute minimum point x0 I for f on I. Show that x0 is a solution to the equation cos x = x2.
-
State the steps in the audit program for the tests of transactions in a manual system that may involve the use of a random sample.
-
Graham Potato Company has projected sales of $6,000 in September, $10,000 in October, $16,000 in November, and $12,000 in December. Of the companys sales, 20 percent are paid for by cash and 80...
-
In the figure an electron (e) is to be released from rest on the central axis of a uniformly charged disk of radius R. The surface charge density on the disk is +4.27 C/m. What is the magnitude of...
-
Martin Manufacturing is preparing its master budget for the first quarter of the upcoming year. The following data pertain to Martin Manufacturing's operations: Current Assets as of December 31...
-
Name and assign R or S stereochemistry to the product(s) you would obtain by reaction of the following substance with ethyl magnesium bromide. Is the product chiral? Is it optically active? Explain.
-
Draw and name the eight isomeric alcohols with formula C5H12O.
-
Roll four six-sided dice ten times, each time recording the sum of the four numbers rolled. Calculate the mean and median of your ten rolls. Repeat this experiment 20 times. Which of these two...
-
If the 5- and 5.5-year par yields are both at 2%, compute the forward rate from 5 to 5.5 years.
-
Solve for the weighted average cost of capital: 10.60% K = cost of equity capital for a leveraged firm 1/3 8.0% = i = before-tax borrowing cost 40.0% = debt-to-total-market-value ratio = marginal...
-
You are a risk consultant to a financial institution. Your client asks you to respond to the following two questions: 1) What are the two common factors for firms to have large losses? 2) List and...
-
When I analyze the activities ratio: inventory turnover average collection period receivable turnover average payable period total asset turnover fixed asset turnover what is the correct analysis...
-
International Data Systems' information on revenue and costs is relevant only up to a sales volume of 109,000 units. After 109,000 units, the market becomes saturated and the price per unit falls...
-
Kale Wilson, an auditor with Sneed Chartered Accountants, is performing a review of Platinum Stereos inventory account. Platinum did not have a good year, and top management is under pressure to...
-
7. Baladna wants to analyze process that includes delivery by suppliers, production inside the company, transportation to to its customers and information systems. Then it also wants to find out...
-
A unit cell of an ionic crystal (a) Shares some ions with other unit cells; (b) Is the same as the formula unit; (c) Is any portion of the crystal that has a cubic shape; (d) Must contain the same...
-
One compound whose molecular formula is C4H6 is a bicyclic compound. Another compound with the same formula has an infrared absorption at roughly 2250 cm-1 (the bicyclic compound does not). Draw...
-
Which compound would you expect to be the more stable: cis-1,2-dimethylcyclopropane or trans-1,2-dimethylcyclopropane? Explain your answer.
-
Consider that cyclobutane exhibits a puckered geometry. Judge the relative stabilities of the 1,2-disubstituted cyclobutanes and of the 1,3-disubstituted cyclobutanes. (You may find it helpful to...
-
Your Boss calls you to his office and asks a couple of questions that are basically not in sequence. What is the scaling technique used to build a distributed Streaming Analytics system such as...
-
Database systems cannot stand alone; they depend on many other systems. Choose an industry from e-commerce, healthcare, or banking and discuss database security for an organization in one of those...
-
Suppose you have this query SELECT Pname, Price, Color FROM PRODUCT WHERE Price < 50 OR Color = Red; Which technique can be used to improve this query ? Q2.With a neat state transition diagram...

Study smarter with the SolutionInn App


