Give IUPAC names (or the following structures: (a) (b) (c) -- CH- H (d) (e)
Question:
Give IUPAC names (or the following structures:
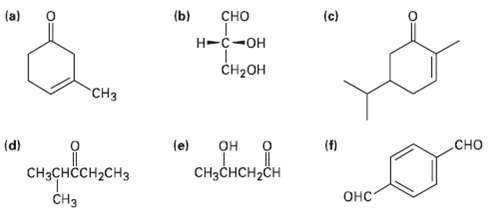
Transcribed Image Text:
(a) (b) сно (c) Н-с-он CH-он "СHз сно (d) (e) (f) он CнзснсH-CH CHзснссH2сHз Онс" Cнз O:
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (18 reviews)
a CH3 3Methyl3cyclo hexenone d b CHCHCH2CH ...View the full answer

Answered By

OTIENO OBADO
I have a vast experience in teaching, mentoring and tutoring. I handle student concerns diligently and my academic background is undeniably aesthetic
4.30+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Give the IUPAC names of the following alkanes. (a) CH3C(CH3)2CH(CH)CH3)CH2CH2CH(CH3)2 (b) (c) (d) (e) (f) (g) (h) CH,CH CHCH CH, CH CH CH,CHCH CH,CHCH CH,CH CH,CH, CH, CH,CH, CH,CH,CH, C(CH,CH),...
-
Give IUPAC names for the following alkenes: (a) (b) (c) (d) (e) (f) OH CI
-
Give IUPAC names for the following compounds. (a) (b) (c) (d) (e) (f) Ph CH3C C CH CH H3C CH3 - (CH3)3C C-CH (CH3)CH2CH3 CH CHC CC-OH CH,CH CH C-C CH
-
Read the article Problem at INSPEECH and answer the question: Why did Inspeech experience a meltdown in 1988?
-
Refer to "Forces Resisting Change". Which of these factors are most strongly affecting Rita and Juan? Support your answer with examples from the scenario.
-
A heavy-duty flashlight mirror has a parabolic cross section with diameter 6 in. and depth 1 in. a) Position a coordinate system with the origin at the vertex and the x-axis on the parabola's axis of...
-
What are the six primary system development process models?
-
Assume Weithorn & Wesley, the accounting firm, advises Lakeside Seafood that its financial statements must be changed to conform to GAAP. At December 31, 2014, Lakesides accounts include the...
-
Consider the same mechanism again, with m=2.5 kg , d=585 mm , k=200 N/m , and s=1.72 . What is the maximum angular velocity allowable if the collar is to remain at a constant distance r=2600 mm from...
-
Thinkabout the system that handles student admissionsat your university. The primary function of the systemshould be able to track a student from the request forinformation through the admissions...
-
Draw and name the seven aldehydes and ketones with the formula C5H10O. Which are chiral?
-
Give structures that fit the following descriptions: (a) An , -un-saturated ketone, C6H8O (b) An -dike tone (c) An aromatic ketone, C9H10O (d) A diene aldehyde, C7H8O
-
In a qualitative research project, how large should the sample be? How many focus groups, individual depth interviews (IDIs), or online bulletin board focus groups are needed? One suggested rule is...
-
As a skilled economic consultant, you are concerned about the forecast provided in the previous module, based on nominal values. Additional research indicates that the implicit price deflator was...
-
You've been provided the demand and supply equation for a Sydney transport company. P = 300e-0.2Qa P = 2e0.80s Where P, Qa, and Qs denote the price and quantity demanded and quantity supplied,...
-
During COVID, the government provided low-income families with a child tax credit. That policy had a major impact on child poverty in the United States. Now that the credit is going away, child...
-
Mrs Kebede S, consumes two good A and B, when Ke bede,s income increase from birr 4000 to 5000, the quantity of good A demanded by increase Kebede e by 35%. What conclusion do you draw from this.
-
es Determine the ending balance of each of the following T-accounts. Accounts Payable Cash 140 90 2,400 340 100 3,300 60 540 10 Supplies 10,400 1,500 4,200 9,200 Accounts Receivable 800 190 190 190...
-
As we explicitly calculated the path integral of the harmonic oscillator in the previous chapter, it is useful to calculate the partition function for the harmonic oscillator to compare the explicit...
-
Compare and contrast debt financing and equity financing as ways of starting a new business. Does one have an overall advantage over the other? What situation is more favorable to the use of debt...
-
Use bond enthalpies from Table 10.3 to determine whether CH 4 (g), CH 3 OH(g), H 2 CO(g), or HCOOH(g) produces the most energy per gram when burned completely in O 2 (g) to give CO 2 (g) and H 2...
-
Compound A has the molecular formula C6H12O3 and shows a strong IR absorption peak at 1710 cm-1. When treated with iodine in aqueous sodium hydroxide, A gives a yellow precipitate. When A is treated...
-
The following is an example of a reaction sequence developed by Derin C. D'Amico and Michael E. Jung (UCLA) that results in enantiospecific formation of two new chirality centers and a carbon-carbon...
-
Additional evidence for the halogenation mechanisms that we just presented comes from the following facts: (a) Optically active 2-methyl-1-phenylbutan-1-one undergoes acid-catalyzed racemization at a...
-
What is the yield to maturity of a 23-year bond that pays a coupon rate of 8.25% per year, has a $1,000 par value , and is currently priced at $1,298.05?
-
Suppose the exchange rate between U.S. dollars and Swiss francs is SF 1.112 = $1.00, and the exchange rate between the U.S. dollar and the euro is $1.00 = 0.9842 euros. What is the cross-rate of...
-
Use the following information for the Lowell, Inc. for this and the next two questions. Sales $200,000 Debt 95,000 Dividends 5,000 Equity 40,000 Net income 16,000 1.What is the company's...

Study smarter with the SolutionInn App


