Give the products expected (if any) when nitrobenzene reacts under the following conditions. (a) Cl2, FeCl3, heat
Question:
Give the products expected (if any) when nitrobenzene reacts under the following conditions.
(a) Cl2, FeCl3, heat
(b) Fuming HNO3, H2SO4
(c)
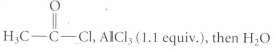
Transcribed Image Text:
H3C-C-CI, AICI,( 1.1 equiv.), then H2O
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (12 reviews)
a b c No electrophilic aromatic substitution reaction ta...View the full answer

Answered By

Guna Perumal
Praveen Mishra has completed his undergraduate degree in Mathematics and also holds a diploma in Electronics Engineering. His educational background reflects a diverse set of skills and knowledge in both mathematical and technical domains. With this combination of education, Praveen is well-equipped to pursue various career opportunities that require a blend of analytical and technical expertise.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Give the products expected when the following compounds are ozonized and reduced. (a) (b) (c) (d)
-
Give the products expected from the following reactions. (a) Acetyl chloride + ethylamine (b) (c) (CHs2NIH benzoyl chloride dimethylamine NH CH3 (CH24-C -CI+ hexanoyl chloride piperidine
-
Give the products expected when the following tertiary amines are treated with a peroxyacid and heated. (a) N, N-dimethylhexan-2-amine (b) N, N-diethylhexan-2-amine (c) Cyclohexyldimethylamine (d)...
-
Shalom Company projects the following operating results next year: Produced 70,000 units Sales 60,000 units Sales price 10 per unit Total fixed cost 300,000 per year Total variable cost 20% of sales...
-
On January 1, 2013, Cook Textiles leased a building with two acres of land from Peck Development. The lease is for 10 years at which time Cook has an option to purchase the property for $100,000. The...
-
Record the transactions of Joan Arendse in Question 6.8 into the general journal. Data from Question 6.8. On 1 March, Joan Arendse (an accounting consultant) had these assets and liabilities:...
-
What are the symptoms of groupthink and how can team leaders prevent it from happening?
-
Athletic Universe began January with merchandise inventory of 65 crates of vitamins that cost a total of $3,835. During the month, Athletic Universe purchased and sold merchandise on account as...
-
Scalia Systems manufactures rugged handheld computers for use in adverse working environments. Scalia tries to maintain inventory at 40% of the following month's expected unit sales. Scalia began the...
-
The following transactions of Carswell Wholesale Inc. occurred in the month of September 2016: Date: Sept. 1Issued 200 common shares for $20,000. 4 To raise additional capital, Carswell borrowed...
-
Using benzene and any other reagents, outline a synthesis of each of the following compounds. Tert-butylcyclohexane
-
Explain how you would distinguish between ethylbenzene, p-xylene, and styrene solely by NMR spectroscopy.
-
This ethical dilemma highlights the tension that can occur between shareholder priorities and social responsibility using an example from Oh Seas!, Co., a cruise line company that deals exclusively...
-
Alvin has lived with his girlfriend, Angel, & her child in his home since July 2022. Angel had earned income of $5,000 and is not required to file a tax return. Alvin provides more than half of their...
-
Your fixed cost is $524,000, your sales price is $1,750/unit, and your variable cost is $938/unit. If you lower the sales price to $1,700, what impact will that have on the number of units must you...
-
If you could buy an investment now and four years later sell it for $27,000, what would you be willing to buy it for, assuming a 5% discount rate and no other cash flows?
-
How does limited liability partnership differ from a general partnership in terms of liability? Discuss in details.
-
If the annual interest rate is 8%, what would you expect to pay for a bond paying a lump sum of $10,000 in ten years? Show your calculations.
-
An element on the surface of a drive shaft is in pure shear and is subjected to stresses xy = 2700 psi, as shown in the figure. Using Mohrs circle, determine the following. (a) The stresses acting...
-
Evaluate the line integral, where C is the given curve. C x 2 dx + y 2 dy, C consists of the arc of the circle x 2 + y 2 = 4 from (2, 0) to (0, 2) followed by the line segment from (0, 2) to (4, 3)
-
Provide both common and IUPAC systematic names for the following thioester: O C. S
-
The G for ATP hydrolysis is 230.5 kJ mol 1 (27.3 kcal mol 1 ). In the red blood cell at 37C (310 K) the concentrations of ATP, ADP, and phosphate are 2.25, 0.25, and 1.65 mM, respectively. Show that...
-
The proton NMR of the C1 proton region of D-glucopyranose shows that both anomers are present. (The large peak in the middle is residual HDO in the H 2 O solvent.) The integrals are given in...
-
The hawkins company maintains one set of financial records for financial reporting purpose. separate records are also kept for tax compliance purpose. why is that necessary?
-
Managerial accounting must follow GAAP. Financial accounting does not. There is no significant difference between financial and managerial accounting. Managerial accounting provides information for...
-
Troy Abraham had the following assets and liabilities as of October 1, 2019. Assets Liabilities Cash $6,500 Automobile $8,700House $165,000 Contents of Home $6,400 Prepaid Insurance $1,700 Unpaid...

Study smarter with the SolutionInn App


