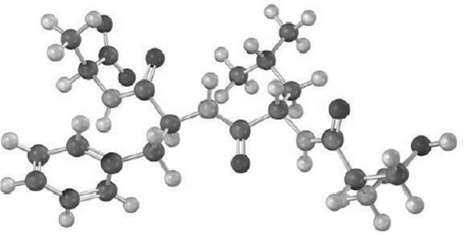
Give the sequence of the following tetrapeptide:
Question:
Give the sequence of the following tetrapeptide:
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (9 reviews)
After identifying the amino acid residues notice that the te...View the full answer

Answered By

CHARLES AMBILA
I am an experienced tutor with more than 7 years of experience. I have helped thousands of students pursue their academic goals. My primary objective as a tutor is to ensure that students have easy time handling their academic tasks.
5.00+
109+ Reviews
323+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Give the sequence of the following tetra peptide (yellow =S):
-
Give an example of a divergent sequence {an} such that converges. lim anl
-
Give a simple example of a page reference sequence where the first page selected for replacement will be different for the clock and LRU page replacement algorithms. Assume that a process is...
-
Explain why entrepreneurial firms are often in a strong position to use combination strategies.
-
Amateur and professional golfers in search of lower scores want to find clubs that are optimized for their individual swings. This process of club fitting has gone decidedly high tech in recent...
-
Although nitration of pyridine requires a temperature of 300°C (eq. 13.2), 2,6 dimethylpyridine is readily nitrated at 100°C. Write an equation for the reaction, and explain why milder...
-
A heat exchange system is needed to cool \(60,000 \mathrm{lb} / \mathrm{hr}\) of acetone at \(250^{\circ} \mathrm{F}\) and 150 psia to \(100^{\circ} \mathrm{F}\). The cooling can be achieved by...
-
The records of Grade A Steak Company list the following selected accounts for the quarter ended April 30, 2012: Requirements 1. Prepare a multi-step income statement. 2. M. Davidson, manager of the...
-
Everything Is Not What It Seems You are a perinatal unit coordinator at a large teaching hospital. In addition to your management responsibilities, you have been asked to fill in as a member of the...
-
Enhance the Treasury Yield Rates database file to perform a database query that finds the rate associated with any date and term.
-
Is the following structure a D amino acid or an L amino acid? Identifyit.
-
Except for cysteine, only S amino acids occur in proteins. Several R amino acids are also found in nature, however. (R)-Serine is found in earthworms, and (R)-alanine is found in insect larvae. Draw...
-
Pam Duffy is reviewing the principle of segregation of duties. What are the two common applications of this principle?
-
Provide TWO big brands/companies you think a person who has a passion for home renovation and construction/ ready mix concrete industry (production line, operations and sales) could be an influencer...
-
Health Care Information Standards and Structures Question How can public health agencies that utilize health information exchanges make their patients feel more comfortable with consenting/...
-
Vital-E Nutrition and answer the following: What types of track and trace capabilities could help VEN detect temperature problems and respond before the product is ruined? Are autonomous trucks a...
-
Q5. Meiying is a student studying Finance Law. Meiying has been watching the stock markets all her life and finds them extremely interesting. Meiying has developed a good sense of the market and has...
-
Kevin wanted to make some extra money to help pay for his educational expenses. As he was looking through the classified ads, he saw a job paying $15.00 per hour for part-time bathroom attendant at a...
-
In Problem 126, if Juan decided to wait 10 years before investing for retirement, how much would he have to invest on his thirty-sixth birthday to have the same account balance on his sixtieth...
-
Calculate the Lagrange polynomial P 2 (x) for the values (1.00) = 1.0000, (1.02) = 0.9888, (1.04) = 0.9784 of the gamma function [(24) in App. A3.1] and from it approximations of (1.01) and (1.03).
-
Based on their molecular structure, pick the stronger acid from each pair of oxyacids. Explain your choice. a. HSO4 or HSO3 c. HCIO or HBrO b. HClO or HCIO d. CC13COOH or CH3COOH
-
a. Name (CH3)2CHCH2CONH2 b. Write the structure of 1-phenylcyclopentanecarboxamide
-
Show that hydrogen bonding is possible for acetamide, but not for N,N-dimethylacetamide.
-
Using eq. 10.42 as a model, write an equation for the hydrolysis of acetamide. R-C-NH.+H-C-OH +NH,(10.42) H+ or HOT amide acid
-
How do Software-Defined Networking (SDN) and Network Functions Virtualization (NFV) paradigms transform traditional network architecture, and what challenges might arise when implementing these...
-
The pension fund you manage must make a payment of $250,000 in ten years. The ten-year interest rate is currently 8% per annum. You want to fund this liability using five-year zero-coupon bonds and...
-
A 1-year zero coupon bond has a YTM of 10%. A 2-year zero coupon bond has a YTM of 12%. Both are riskless. What is the price of a 2-year, annual 5% coupon paying bond with face value of $1,000?

Study smarter with the SolutionInn App


