Based on their molecular structure, pick the stronger acid from each pair of oxyacids. Explain your choice.
Question:
Based on their molecular structure, pick the stronger acid from each pair of oxyacids. Explain your choice.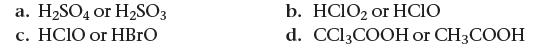
Transcribed Image Text:
a. H₂SO4 or H₂SO3 c. HCIO or HBrO b. HClO₂ or HCIO d. CC13COOH or CH3COOH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (2 reviews)
a HSO4 more oxygen atoms bonded ...View the full answer

Answered By

Emily Grace
With over a decade of experience providing top-notch study assistance to students globally, I am dedicated to ensuring their academic success. My passion is to deliver original, high-quality assignments with fast turnaround times, always striving to exceed their expectations.
4.90+
3+ Reviews
22+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Based on their molecular structure, pick the stronger acid from each pair of binary acids. Explain your choice. a. HF and HCI b. HO or HF c. HSe or HS
-
Based on their molecular structure, pick the stronger acid from each pair of oxyacids. Explain your choice. MISSED THIS? Read Section 17.10 a. HSO4 or HSO3 c. HCIO or HBrO b. HClO or HCIO d. CCI,COOH...
-
Sam's Insurance must choose between two types of printers. Both printers meet the firm's quality standard. Printer A costs $3,500 and is expected to last 3 years with operating costs of $380 per...
-
For the transfer function below, find the constraints on K 1 and K 2 such that the function will have only two j poles. K1s + K2 T(s) = s4 + K1s + s? + K2s + 1
-
Consider the following conversation between Keri Swasey, manager of a division that produces riding lawn mowers, and her controller, Stoney Lawson, a CMA and CPA: Keri: Stoney, we have a real...
-
Berkley Corporation issued bonds and received cash in full for the issue price. The bonds were dated and issued on January 1, 2009. The stated interest rate was payable at the end of each year. The...
-
Search for a California case that lists the essential elements of a cause of action for a particular contract case. If your library has Witkins Summary of the Law, a multivolume treatise that...
-
Following are audit procedures that are normally conducted in the purchasing process and related accounts. 1. Test a sample of purchase requisitions for proper authorization. 2. Test transactions...
-
1. Was the removal of the Multi-Fiber Agreement a positive thing for the world economy? Why? 2. As a producer in a developing nation such as Bangladesh that benefited from the MFA agreement. how...
-
Based on molecular structure, arrange the oxyacids in order of increasing acid strength. Explain your choice. HClO3, HIO3, HBrO3
-
Based on molecular structure, arrange the binary compounds in order of increasing acid strength. Explain your choice. HTe, HI, HS, NaH
-
Examine the function for relative extrema and saddle points. f(x, y) = 2xy (x + y) + 1 - + Z -2 2 3 X
-
Calculate the Black-Scholes Values for a call option and a put option. Share price (S): $100 Exercise price (X): $95 Continuous Risk-free Interest rate (r): 8% Dividend yield (q): 3% Time to Maturity...
-
What are Enterprise Environmental Factors ( EEFs ) in the context of procurement management?
-
It is December 2022. Josephine Cochrane owns and operates a small-appliance repair shop. The shop rents space in a building and is quite busy. Ms. Cochrane is considering constructing a new. larger...
-
Use the Present Value of $1 table to determine the present value of $1 What is the total present value of the cash flows received received one year from now? Assume a 6% interest rate. Present Value...
-
What is the initial investment for a Straddle with a strike price of 200? Price Today (So) 202.47 Call Premium Put Premium Strike Bid Ask Bid Ask 190 23.85 26.30 11.60 13.25 200 17.95 20.20 15.80...
-
Why do creditors usually accept a plan for financial rehabilitation rather than demand liquidation of a business?
-
Assume Eq. 6-14 gives the drag force on a pilot plus ejection seat just after they are ejected from a plane traveling horizontally at 1300 km/h. Assume also that the mass of the seat is equal to the...
-
Using Eq. (6.2), derive an expression for the focal length of a homogeneous transparent sphere of radius R. Locate its principal points. (n 1)d (n 1) R1 (6.2) R2 nRR2
-
A spherical glass bottle 20 cm in diameter with walls that are negligibly thin is filled with water. The bottle is sitting on the back seat of a car on a nice sunny day. Whats its focal length?
-
A thick glass lens of index 1.50 has radii of +23 cm and +20 cm, so that both vertices are to the left of the corresponding centers of curvature. Given that the thickness is 9.0 cm, find the focal...
-
what ways does the protagonist's internal conflict serve as a microcosm for the societal upheaval depicted throughout the novel ?
-
Evaluate the efficacy of the author's experimental narrative form in conveying the fragmented nature of memory and trauma within the context of historical fiction .
-
Britta Jones is a widowed mother of two who lives on Harrison Boulevard in the Historic District in Boise, Idaho. Britta hired Steve to help run the Jones family household. This full-time, live-in...

Study smarter with the SolutionInn App


