How could you prepare the following Cyclohexanone by combining a Stork enamine reaction with an intra molecular
Question:
How could you prepare the following Cyclohexanone by combining a Stork enamine reaction with an intra molecular aldol condensation?
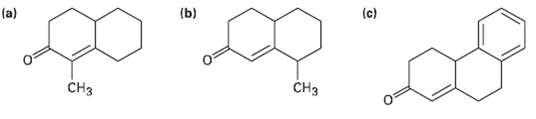
Transcribed Image Text:
(b) (c) (a) CHз CHз
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 64% (14 reviews)
a CH3CHCCHCH 00100 Michael enamine formation CH3 HO heat dehydrati...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The following reaction involves an intra molecular aldol reaction followed by a ret ro aldol-like reaction. Write both steps, and show theirmechanisms. O2Et CO2Et Na* "OEt Ethanol
-
How could you prepare the following compounds with benzene as one of the starting materials? (a) (b)
-
a. Starting with isopropylacetylene, how could you prepare the following alcohols? 1. 2-methyl-2-pentanol 2. 4-methyl-2-pentanol b. In each case a second alcohol would also be obtained. What alcohol...
-
In the conversion of a-ketoglutarate to glucose, which of the following compounds are not obligatory intermediates in this pathway? * Oxaloacetate O Fructose 1,6 bisphosphate O Malate O 1,3...
-
How might you perform "risk adjustment" of Consumer Reports automobile ratings?
-
Some ions do not have a corresponding neutral atom that has the same electron configuration. For each of the following ions, identify the neutral atom that has the same number of electrons and...
-
What is the purpose of using the BSC in the IT strategic planning process?
-
Information pertaining to Hedlund Corporation's property, plant, and equipment for 2011 follows. The salvage value s of the depreciable assets are immaterial. Depreciation is computed to the nearest...
-
1) Some people complain about being poor. Others blame their parents, families, governments and others for their condition. However, wealth seems to be related to productivity. More productive people...
-
In addition to carrying goods from one place to another, some trucking companies provide supply chain management services and help their customers manage their information. In this project, youll use...
-
The Stork en amine reaction and the intra molecular aldol reaction can be carried out in sequence to allow the synthesis of Cyclohexanone. For example, reaction of the pyrrolidine enamine of...
-
The amino acid leucine is biosynthesized from a-ketoisovalerate by the following sequence of steps. Show the mechanism ofeach. Acetyl CoA COASH NAD+ NADH/H* CO2 "C 2-Isopropylmalate co2 ...
-
Match each of the following items A through G with the best numbered description of its purpose. A. Raw Materials Inventory account B. Materials requisition C. Finished Goods Inventory account D....
-
Laramie Company has variable cost ratio of 0 . 3 5 . The fixed cost is $ 1 1 3 , 7 5 0 and 2 5 , 0 0 0 units are sold at break - even. What is the price? What is the variable cost per unit? The...
-
Pension plan assets were $ 4 6 0 million at the beginning of the year and $ 4 8 7 million at the end of the year. The return on plan assets was 5 % . At the end of the year, cash invested in the...
-
KJ Company received a piece of land at market value for $80,000. The asking price was $90,000. Through this transaction, 30,000 common shares were issued with a par value of $1 per share. Prepare the...
-
On January 1, Dora purchases 175 of the $1,000, 7%, 15-year bonds issued by Splash City, with interest receivable semiannually on June 30 and December 31 each year. Assuming the market interest rate...
-
On January 1, 2012, Adam Corp signed a 5-year non-cancellable lease for one (non-specialized) machine. The terms of this contract require annual payments of $9,968 at the beginning of each year...
-
Global warming might result in the melting of the polar ice caps. What effect would this melting have on the length of the day?
-
Data on weekday exercise time for 20 females, consistent with summary quantities given in the paper An Ecological Momentary Assessment of the Physical Activity and Sedentary Behaviour Patterns of...
-
Use covalent Lewis structures to explain why the compound that forms between nitrogen and hydrogen has the formula NH 3 . Show why NH 2 and NH 4 are not stable. 10.5 Covalent Bonding: Lewis...
-
Propose mechanisms and predict the major products of the following reactions. Include stereochemistry where appropriate. (a) cycloheptene + Br2 in CH2Cl2 (c) (E)-dec-3-ene + Br2 in CCl4 (d)...
-
Propose a mechanism for the addition of bromine water to cyclopentene, being careful to show why the trans product results and how both enantiomers are formed.
-
(a) When 1 mol of buta-1,3-diene reacts with 1 mol of HBr, both 3-bromobut-1-ene and 1-bromobut-2-ene are formed. Propose a mechanism to account for this mixture of products. (b) When...
-
Agencies have been entrusted with a vast amount of power to carry out their missions. Social changes can quickly develop as a result of changes in laws or public policy. During times of rapid changes...
-
As manager of a hotel, you have noticed that persons suspected to be prostitutes are seen frequently in your hotel lobby and bar. What are your options, considering legal issues and business...
-
Bakrah El Hib, aged 61, was driving to work on 2nd August when he was struck by Yunis Khallah's car at the Wallchild roundabout at 8:30 am. As a result of the summer school holidays, the roundabout...

Study smarter with the SolutionInn App


