The Stork en amine reaction and the intra molecular aldol reaction can be carried out in sequence
Question:
The Stork en amine reaction and the intra molecular aldol reaction can be carried out in sequence to allow the synthesis of Cyclohexanone. For example, reaction of the pyrrolidine enamine of Cyclohexanone with 3-huten-2-one, followed by enamine hydrolysis and base treatment, yields the product indicated. Write each step, and show the mechanism ofeach.
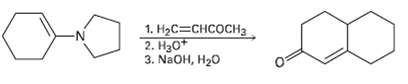
Transcribed Image Text:
1. НаС%3снсосHз 2. Нао+ 3. NaOH, H20 -N
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 46% (15 reviews)
HCCHCCH3 1 HO 2 heat Michael add...View the full answer

Answered By

ALBANUS MUTUKU
If you are looking for exceptional academic and non-academic work feel free to consider my expertise and you will not regret. I have enough experience working in the freelancing industry hence the unmistakable quality service delivery
4.70+
178+ Reviews
335+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The production of 1,3-butadiene can be carried out by the dehydrogenation of 1- butene: C2H5CH:CH2(g) CH2:CHCH:CH2(g) + H2(g) Side reactions are suppressed by the introduction of steam. If...
-
The production of 1,3-butadiene can be carried out by the dehydrogenation of n- butane: C4H 10(g) CH2 : CHCH : CH2(g) + 2H2(g) Side reactions are suppressed by the introduction of steam. If...
-
This activity is a project that can be carried out in a team of two students. It provides an exercise in searching for data and building a model using the U.S. Census data (census.gov). Check for the...
-
Select the best answer. 1. A local chapter of the Society for Protection of the Environment beneted from the voluntary services of two attorneys. One served as a member of the Society's board of...
-
"An analysis of sustainability is similar to a five-force analysis." Comment.
-
The discovery of hafnium, element number 72, provided a controversial episode in chemistry. G. Urbain, a French chemist, claimed in 1911 to have isolated an element number 72 from a sample of rare...
-
What are the four BSC perspectives?
-
FINANCIAL STATEMENTS Page 203 shows a work sheet for Monte's Repairs. No additional investments were made by the owner during the month. REQUIRED 1. Prepare an income statement. 2. Prepare a...
-
How can Southeast Groceries Leadership encourage others to embrace and support change when rumors are flying? If you were head of HR at SEG, what would be your communication plan to employees for...
-
The Britts & Straggon company manufactures small engines at three different plants. From the plants, the engines are transported to two different warehouse facilities before being distributed to...
-
When 2-methylcyclohexanone is converted into an enamine, only one product is formed despite the fact that the starting ketone is unsymmetrical. Build molecular models of the two possible products,...
-
How could you prepare the following Cyclohexanone by combining a Stork enamine reaction with an intra molecular aldol condensation? (b) (c) (a) CH CH
-
How important are each of the following considerations in an ERP software? a. Ease of use b. Complete integration c. Data reliability
-
Assuming that the perpetual inventory method is used and costs are computed at the time of each withdrawal, what is the gross profit if the inventory is valued at FIFO?
-
Tesla purchased an auto-piloting system for $10,000,000 on January 1, 2016. It expects to use the system for 5 years, estimates a salvage value of $500,000, and uses the straight-line deppreciation n...
-
Calculate the retained earnings on December 31, 2025? Sheridan Supply Company compiled the following financial information as of December 31, 2025: Service revenue $1156000 Common stock 236000...
-
In September, Lauren Ashley Company purchased materials costing $220,000 and incurred direct labor cost of $150,000. Overhead totaled $300,000 for the month. Information on inventories was as...
-
Analyze the accounting issues raised by the Enron case. If you knew about these facts before they became public, what, if anything, would you have done? What does this case demonstrate about the...
-
The structure in Figure P11.60 is made of three identical rods of uniform density. Rank the rotational inertias of the structure, from smallest to largest, about the four rotation axes represented by...
-
How does the organizational structure of an MNC influence its strategy implementation?
-
Write the electron configuration for Ne. Then write the Lewis symbol for Ne and show which electrons from the electron configuration are included in the Lewis symbol.
-
Show how you would accomplish the following transformations. (a) (b) (c) 1-methylcycloheptanol 2-methylcycloheptanol
-
(a) When HBr adds across the double bond of 1,2-dimethylcyclopentene, the product is a mixture of the cis and trans isomers. Show why this addition is not stereospecific. (b) When...
-
Give mechanisms to account for the stereochemistry of the products observed from the addition of bromine to cis- and trans-but-2-ene (Figure 8-4). Why are two products formed from the cis isomer but...
-
Ala was prosecuted for murder of his friend Sam. Ala admitted that they had argued and that he had punched Sam, knocking him down. However, when testifying in his own defence, he claimed that the...
-
Going back to this week's recording, revise what you learnt about the doctrine of precedent. In the famous House of Lords case of Donoghue v Stevenson [1932] AC 562, the Court determined that the...
-
Joker invites Batman and Robin to his home for tea. Peter Paralegal who is also attending the meeting, presents a will he prepared. He asks Batman and Robin to be physically present when Joker signs...

Study smarter with the SolutionInn App


