How might the following amines be prepared using reductive amination reactions? Show all precursors if more than
Question:
How might the following amines be prepared using reductive amination reactions? Show all precursors if more than one ispossible.
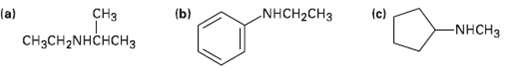
Transcribed Image Text:
CHз (b) NHCH2CH3 (c) (a) CHзCH2NHCHсHз -NHCH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 53% (13 reviews)
Look at the target molecule to find the groups bonded to nitrogen One group ...View the full answer

Answered By

Branice Buyengo Ajevi
I have been teaching for the last 5 years which has strengthened my interaction with students of different level.
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
How might the following compounds be prepared using Michael reactions? Show (he nucleophilic donor and the electrophilic acceptor in eachcase. ", , (b) . (a) CHCCH-CH2CH2CH3 CH2H2CCgHs o2Et NO2 (d)...
-
How might the following business specialists use learning curves: accountants, marketers, financial analysts, personnel managers, and computer programmers?
-
How might the following influences affect a firms business risk (consider each separately)? a. Imports increase the level of competition b. Labor costs decline c. Health care costs (provided for all...
-
DRAW A PLAN OF YOUR HOME THEN ESTIMATE QUANTITY OF * FOOTING *BLOCK WALL UNDER DPC * BLOCK WALL ABOVE DPC * CEMENT PLASTERING OUTSIDE *GYPSUM PLASTERING INSIDE
-
How can organizations motivate employees to promote safety and health in the workplace?
-
Cite three metallurgical/processing techniques that are employed to enhance the creep resistance of metal alloys.
-
To fit the quadratic regression model using MINITAB, when the \(x\) values are in C1 and the \(y\) values in \(\mathrm{C} 2\), you must select Use the computer to repeat the analysis of Example 11....
-
The scheduled arrival time for a daily l ight from Boston to New York is 9: 25 am. Historical data show that the arrival time follows the continuous uniform distribution with an early arrival time of...
-
Explain the importance of water sustainability in the broader context of environmental stewardship, addressing the challenges of water scarcity, water quality, and water-related risks, and examining...
-
Use the following forward and spot prices for Canadian dollars (C$) to answer the question below. The prices are in U.S. dollars ($/C$). Forward rate for 4/15/20 Spot rate delivery of Canadian...
-
Show two methods for the synthesis of dopamine, a neurotransmitter involved in regulation of the central nervous system. Use any alkyl halideneeded. Dopamine
-
How could you prepare the following amine using a reductive aminationreaction?
-
Texas Roadhouse opened a new restaurant in October. During its first three months of operation, the restaurant sold gift cards in various amounts totaling $2,500. The cards are redeemable for meals...
-
Coverage of key literature related to this research topic and organizing it in a well-structured way. Critically summarize and link with your research question. Data and method: Also, cover the...
-
2) Find general solutions of the following differential equations: (a) (b) dy+6+9y=5 sinx dx dx d'y dx 4dy +12y=4e-x dx
-
Let us assume that you want to start a business in another country or you work with a corporation that wants to do business in another country. What are some money market tools and/or financial...
-
Consider the following data: Assets E(r) A B C 0 0.05 0.10 0.10 0.20 0.15 0.30 The risk-free rate is r = 0.035. The correlation matrix for the assets is given by: Correlation matrix: (for risky...
-
Frankie bought a trip on her credit card on May 28 for $2,231.00. She paid off the entire amount of the trip including the interest charges 128 days later. The annual interest rate on purchases is...
-
Med Diagnostics Inc. borrowed \($200\),000 from a lender for a new blood analyzer module to improve accuracy and consistency of its tests. The rate was 6 percent, 2 percent above the prime rate. The...
-
A red card is illuminated by red light. What color will the card appear? What if its illuminated by blue light?
-
An equilibrium mixture contains N 2 O 4 (P = 0.28 atm) and NO 2 (P = 1.1 atm) at 350 K. The volume of the container is doubled at constant temperature. Write a balanced chemical equation for the...
-
Suggest which of the following monomers might polymerize well on treatment with BF3. (a) Vinyl chloride (b) Vinyl acetate (c) Methyl a-cyanoacrylate
-
Chain branching occurs in cationic polymerization much as it does in free-radical polymerization. Propose a mechanism to show how branching occurs in the cationic polymerization of styrene. Suggest...
-
Draw the important resonance forms of the stabilized anion formed in the anionic polymerization of methyl acrylate.
-
If you weigh your portfolio in 80% risky and 20% risk-free t bills. The optimal risky portfolio consists of 40% in bonds and 60% in ETFs. How much will you have invested in t-bills, bonds, and etfs?
-
b. A 25-year mortgage loan of $184,000 at a 10 percent compound annual interest rate with equal installment payments at the end of each year.
-
What are Cloud-based Services? How does this differ to the common virtualization model?

Study smarter with the SolutionInn App


