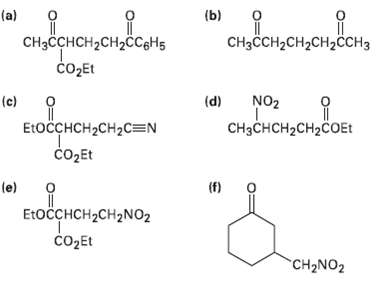
How might the following compounds be prepared using Michael reactions? Show (he nucleophilic donor and the electrophilic
Question:
How might the following compounds be prepared using Michael reactions? Show (he nucleophilic donor and the electrophilic acceptor in eachcase.
Transcribed Image Text:
"ндо, Понененает, (b) Дененене. (a) CHзCCH-CH2CH2CСH3 CнзсснсH2сH2CCgHs čo2Et NO2 (d) (c) П E:OCCHCH2CH2C=N CHзснсH2сH2cOEt СоДEt (П) (e) EtoсснCH2CH2NO2 COДEL CH2NO2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (9 reviews)
Michael reactions occur between stabilized enolate anions and ounsaturated carbonyl compounds Learn ...View the full answer

Answered By

Jonas Araujo
I have recently received the degree of PhD. In Physics by the Universidade Federal do Maranhão after spending a term in Durham University, as I have been awarded a scholarship from a Brazilian mobility program. During my PhD. I have performed research mainly in Theoretical Physics and published works in distinguished Journals (check my ORCID: https://orcid.org/0000-0002-4324-1184).
During my BSc. I have been awarded a scholarship to study for a year in the University of Evansville, where I have worked in detection-analysis of photon correlations in the the Photonics Laboratory. There I was a tutor in Electromagnetism, Classical Mechanics and Calculus for most of that year (2012).
I am very dedicated, honest and a fast learner, but most of all, I value a job well done.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
How could the following compounds be prepared from cyclohexanone? a. b. c. d. SCH3 ,
-
How could the following compounds be prepared from a carbonyl compound with no carbon-carbon double bonds? a. b. CH CH-CHCCH2CH2CH3 C CH-CH2 CH3
-
How could each of the following compounds be prepared from a ketone and an alkyl halide? a. b. CH,CCH2CH CH-CH2 CH2CHCCH2CH3 CH3
-
Racing Investment Fund 2000 LLC was created in August 2000 to purchase, train, and race thoroughbred horses. The LLC ' s operating agreement provided for 50 membership units to be sold for an initial...
-
In light of the winner's curse, must winning bidders in auctions necessarily "lose" in the sense of paying more than the item is worth? What steps can bidders take to prosper in auctions?
-
Use the de Broglie relationship to determine the wavelengths of the following objects: (a) An 85-kg person skiing at 50 km / hr, (b) A 10.0-g bullet fired at 250 m/s, (c) A lithium atom moving at 2.5...
-
Why would a company want to invest in strategic technologies?
-
Sonnet, Inc., has the following budgeted standards for the month of March 2011: Average selling price per diskette $ 6.00 Total direct material cost per diskette $ 1.50 Direct manufacturing labor...
-
Talk Technologies manufactures capacitors for cellular base stations and other communications applications. The company's July 2022 static budget was based on expected sales of 12,000 units is...
-
Is it possible for a firm to have a high current ratio and still find difficulties in paying its current debt? Explain with illustration.
-
In contrast to the rapid reaction shown in Problem 23.40, ethyl acetoacetate requires a temperature over 150 ?C to undergo the same kind of cleavage reaction. How can you explain the difference in...
-
The so-called Wieland?Miescher ketone is a valuable starting material used in the synthesis of steroid hormones. How might you prepare it from 1, 3-cyclohexanedione? Wieland-Miescher ketone
-
Discuss your feelings about the following statement: Management should always fight hard to keep unions out of their organization.
-
Jake, Sacha, and Brianne own a tour company called Adventure Sports. The partners share profit and losses in a 2:3:5 ratio. After lengthy disagreements among the partners and several unprofitable...
-
Simons Company has $400,000 of bonds outstanding. The unamortized premium is $7,400. If the company redeemed the bonds at 101, what would be the gain or loss on the redemption?
-
Smith, Inc., has the following stockholders' equity accounts as of January 1, 2021: Preferred stock-$120 par, nonvoting and nonparticipating, 8% cumulative dividend Common stock-$10 par value...
-
How do you evaluate and mitigate the risks of software regressions and compatibility issues when applying patches across diverse technology stacks ?
-
Mandy Murphy, owner of Murphy and Co. (Saint John, New Brunswick), gave the following list of assets and liabilities to a public accountant and asked him to prepare a balance sheet for the company as...
-
Two skaters skate toward each other, each moving at \(3.3 \mathrm{~m} / \mathrm{s}\). Their lines of motion are separated by a perpendicular distance of \(2.0 \mathrm{~m}\). Just as they pass each...
-
Subprime loans have higher loss rates than many other types of loans. Explain why lenders offer subprime loans. Describe the characteristics of the typical borrower in a subprime consumer loan.
-
Write the Lewis structure for each molecule or ion. a. CI 4 b. N 2 O c. SiH 4 d. Cl 2 CO
-
Write a balanced equation for each reaction. (a) (b) (c) (d) H SO, heat CH3 CH2CH-CH NaOC(CH3 3 Br Br Nal CHCH CH-CH acetone NaOH, heat CH3 CH CCH3 Br
-
For each of the following molecular formulas, determine the number of elements of unsaturation, and draw three examples. (a) C4H4Cl2 (b) C4H8O (c) C6H8O2 (d) C5H5NO2 (e) C6H3NClBr
-
Show how you would prepare cyclopentene from each compound. (a) Trans-1, 2-dibromocyclopentane (b) Cyclopentanol (c) Cyclopentyl bromide (d) Cyclopentane (not by dehydrogenation)
-
Is China's rapid economic growth, spurred by Deng Xiaoping's reforms in the 80s, sustainable in the long term, or does the current threat of deflation indicate potential risks that could undermine...
-
The market for Big Macs in Canada is initially in equilibrium with supply and demand curves of the usual shape. Whoppers are a close substitute for Big Macs; Fries and Big Macs are complements. Use...
-
The COVID-19 pandemic has adversely affected the lives of people around the world in millions of ways. Due to this severe epidemic, all countries in the world have been affected by all aspects,...

Study smarter with the SolutionInn App


