How would you carry out the following transformations using an organo copper coupling reaction? More than one
Question:
How would you carry out the following transformations using an organo copper coupling reaction? More than one step is required in eachcase.
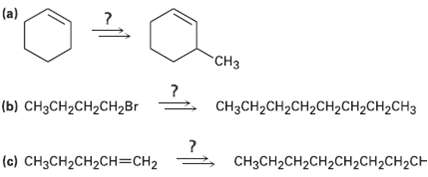
Transcribed Image Text:
(a) "CHз (b) сHзсH2CH2CHгBr CH3CH2CH2CH2CH2CH2CH2CH3 (c) CH3CH2CH2CH=CH2 CHзCH2CH2CH2CH2СH2CH2CH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (15 reviews)
a Strategy The methyl group has an allylic relationship to the double bond Thus an organometallic co...View the full answer

Answered By

Asim farooq
I have done MS finance and expertise in the field of Accounting, finance, cost accounting, security analysis and portfolio management and management, MS office is at my fingertips, I want my client to take advantage of my practical knowledge. I have been mentoring my client on a freelancer website from last two years, Currently I am working in Telecom company as a financial analyst and before that working as an accountant with Pepsi for one year. I also join a nonprofit organization as a finance assistant to my job duties are making payment to client after tax calculation, I have started my professional career from teaching I was teaching to a master's level student for two years in the evening.
My Expert Service
Financial accounting, Financial management, Cost accounting, Human resource management, Business communication and report writing. Financial accounting : • Journal entries • Financial statements including balance sheet, Profit & Loss account, Cash flow statement • Adjustment entries • Ratio analysis • Accounting concepts • Single entry accounting • Double entry accounting • Bills of exchange • Bank reconciliation statements Cost accounting : • Budgeting • Job order costing • Process costing • Cost of goods sold Financial management : • Capital budgeting • Net Present Value (NPV) • Internal Rate of Return (IRR) • Payback period • Discounted cash flows • Financial analysis • Capital assets pricing model • Simple interest, Compound interest & annuities
4.40+
65+ Reviews
86+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
How would you carry out the following transformations? "Co CH2 C Lc
-
How would you carry out the following transformations? Co .CO2H (a) (b) Co CH2 C (c) CH2SH
-
How would you carry out the following reactions to introduce deuterium into organicmolecules? (a) CHH2C3CCH2C Hs 2H5 (b) C2H5 CHH2C3CCH2CH3 C=C Hs D (c) CH3CH2CH2C=CH CH3CH2CH2C=CD CD CD2 C=CH (d)
-
Here are the comparative income statements of Eudaley Corporation. Instructions (a) Prepare a horizontal analysis of the income statement data for Eudaley Corporation, using 2013 as a base. (Show the...
-
Data covering the most recent 30 days are given in the following table for the price per gallon of regular gasoline at a local station. a. Compute a forecast for the next day's price by using a 3-day...
-
A propped cantilever beam of length 2L with support at B is loaded by a uniformly distributed load with intensity q. Use the method of superposition to solve for all reactions. Also draw shear-force...
-
Why is it difficult to implement enterprise systems?
-
Carolyn is senior vice president of finance and chief actuary for Rock Solid Insurance Company (RSIC). Lonnie is double-majoring in finance and mathematics at State University. Lonnie applied for an...
-
The diagram shows the instant when a long slender bar of mass 4.8 kg and length 2.9 m is horizontal. At this instant the mass m= 6.2 kg has a vertical velocity of 5.3 m/s. If the pulley has...
-
Figure 9.36 is the tin-gold phase diagram, for which only single-phase regions are labeled. Specify temperature-composition points at which all eutectics, eutectoids, peritectics, and congruent phase...
-
How might you replace a halogen substituent by a deuterium atom if you wanted to prepare a deuteratedcompound? Br , CHH-H CHCH2CH
-
Rank each of the following series of compounds in order of increasing oxidationlevel: CI (a) (b) CH3CN CH3CH2NH2 H2NCH2CH2NH2
-
Lots A and B were adjacent properties in a residential community near the Chesapeake Bay. Their only access to the beach was through a vacant tract of land the Disputed Area which was not part of...
-
Patrick Star Enterprises has a 15-year bond issue outstanding that pays a 9 percent coupon. The bond is currently priced at $894.60 and has a par value of $1,000. Interest is paid semiannually. What...
-
Manuel Patron Inc. announced a bid to take over Vic Orbison Limited. Manuel Patron Inc. has 3 million shares outstanding, selling at $15 per share. Vic Orbison Limited has 2 million shares...
-
Gingerbread Ltd is an Australian Securities Exchange (ASX) listed company that is currently undergoing an equal access share buy-back scheme. The market price of Gingerbread Ltd is currently $14.50...
-
Firm A has a market value of $600 MM and 30 MM shares outstanding. Firm B has a market value of $200 MM and 20 MM shares outstanding. A is contemplating acquiring B. Firm A's CFO concludes that the...
-
Consider a semiannual 6% coupon bond with a $1,000 face value that has 8 years to maturity. (a) Calculate the market price of this bond using a yield to maturity (YTM) of 5.4%. (b) Is the bond in...
-
According to you, which is the best among on-the-job and off-the-job training?
-
Banner Company acquires an 80% interest in Roller Company for $640,000 cash on January 1, 2013. The NCI has a fair value of $160,000. Any excess of cost over book value is attributed to goodwill. To...
-
Sodium reacts with water according to the reaction: Identify the oxidizing agent. 2 Na(s) + 2 HO(1) 2 NaOH(aq) + H(g)
-
Draw the most stable conformation of: (a) Cis-1-tert-butyl-3-ethylcyclohexane (b) Trans-1-tert-butyl-2-methylcyclohexane (c) Trans-1-tert-butyl-3-(1,1-dimethylpropyl)cyclohexane
-
Write structures for the following compounds. (a) 3-ethyl-4-methylhexane (b) 3-ethyl-5-isobutyl-3-methylnonane (c) 4-tert-butyl-2-methylheptane (d) 5-isopropyl-3, 3, 4-trimethyloctane
-
Name the following compounds. (a) (b) (c) (d)
-
Provide References In September of 2015, the EPA issued Volkswagen ( OTCPK:VLKAY ) a notice of violation of the Clean Air Act of 1963. The EPA accused the Volkswagen Group of intentionally...
-
2. Find c such that A is invertible 1 0 A= -1 -1 3 1 0 2c -4]
-
Futura Company purchases the 4 0 , 0 0 0 starters that it installs in its standard line of farm tractors from a supplier for the price of $ 8 . 4 0 per unit. Due to a reduction in output, the company...

Study smarter with the SolutionInn App


