How would you carry out the following transformations? Co CH2 C Lc
Question:
How would you carry out the following transformations?
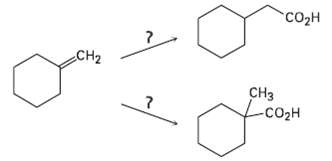
Transcribed Image Text:
"Coон CH2 CНз Lcозн
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (10 reviews)
a CH H CH2OH CHBr PBr3 1 BH3 THF 2 HO OH Grignard carboxylation ...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
How would you carry out the following transformations using an organo copper coupling reaction? More than one step is required in eachcase. (a) "CH (b) HH2CH2CHBr CH3CH2CH2CH2CH2CH2CH2CH3 (c)...
-
How would you carry out the following transformations? More than one step may berequired. H-CH (a) (b) Br - (c) " - " - (d) CH3CH2CH2CH2C=CH CHH2CH2CH2CH2H20CH3 H (e) CH3CH2CH2CH2CHCH3...
-
How would you carry out the following transformation, a step used in the commercial synthesis of(S)-ibuprofen? CN
-
Which do you feel is more important for a new business: the income statement or the statement of ash flow? Why?
-
Ask an experienced leader what he or she thinks is the most effective method of developing leadership skills. Bring your findings back to class.
-
On September 1, Chris Eugene made the following band equipment purchases at Leslie's Music Store. Calculate her total bill. Complete a check for the correct amount. Print a copy of the check from...
-
What is the System Element Architecture (SEA) Construct? What purpose does it serve? Where is it applied?
-
John Dough has just been awarded his degree in business. He has three education loans outstanding. They all mature in 5 years and can be repaid without penalty any time before maturity. The amounts...
-
3. (15 points) Sasha is a well-paid accountant, and for every hour she works, she earns $50 before taxes. Further- more, suppose that she can work up to 10 hours per day, 365 days per year. Draw her...
-
On October 5, Tina Serelio performed legal services for a client who could not pay immediately. The business expected to collect the $12,000 the following month. On November 18, the business received...
-
Using 13 CO 2 as your only source of labeled carbon, along with any other compounds needed, how would you synthesize the following compounds? (a) CH 3 CH 2 13 CO 2 H (b) CH 3 13 CH 2 CO 2 H
-
Which method?Grignard carboxylation or nitrile hydrolysis?would you use for each of the following reactions? Explain. " - C CH2Br (a) " CH Br (b) CH3CH2CHCO2H CCH2CHCH3 (c) CHH-H2H2, CHH-H-CH21...
-
Consider the cart shown in Figure. Suppose we model the wheels as solid disks. Then the wheel inertia is given by IW = mWRw2/2. How small must the wheel mass mw be relative to the cart body mass mc...
-
As of December 31, 2014, Toyota company: had assets of 13,050,000, liabilities of 4,650,000, share capital of 3,300,000 and retained earnings of 5,100,000. What is the Total equity as of that date?
-
What role do serendipitous encounters and unexpected stimuli play in triggering moments of inspiration, and can they be intentionally cultivated ?
-
Oslo Company prepared the following contribution format income statement based on a sales volume of 1,000 units (the relevant range of production is 500 units to 1,500 units): Sales Variable expenses...
-
Public Company acquired the net assets of Sun Company by paying cash $420,000. At that time, the book value of the net assets of Sun Company was $300,000 and the fair value its net assets was...
-
Parker Inc. makes and sells toys. The information on the cost per unit is as hillews. Direct materials $2.55 Direct labor 1.20 Variable overhead 1.90 Variable marketing expense 0.40 The Tooed...
-
In problem 49, when does the estate's taxable year end? problem 49, John Henry died on May 1, 2018. When does his final 1040 have to be filed?
-
Complete the following acid-base reactions: (a) HCCH + NaH
-
Classify each of the following as a component of a silicate ceramic, an oxide ceramic, or a nonoxide ceramic. a. B 4 C b. Mg 2 SiO 4 c. MoSi 2
-
The guanidino group of arginine is one of the most strongly basic of all organic groups. Explain. NH NI NI
-
(a) dl-Glutamic acid has been synthesized from diethyl acetamidomalonate in the following way. Outline the reactions involved. (b) Compound G has also been used to prepare the amino acid dl-ornithine...
-
Synthetic polyglutamic acid exists as an a helix in solution at pH 2-3. When the pH of such a solution is gradually raised through the addition of a base, a dramatic change in optical rotation takes...
-
Assume that management wants wage concession from its counterpart labor union. Explain why this often leads to disputes. Suppose management gives up some of its future negotiating power by subjecting...
-
Why have you chosen this nursing Field of Specialization or Major? What influenced you in selecting your intended major of study and what goal(s) have you set for the future?
-
What are logical fallacies and how can they affect our decision-making?

Study smarter with the SolutionInn App


