How would you prepare the following amines, using both Hofmann and Curtius rearrangements on a carboxylic acidderivative?
Question:
How would you prepare the following amines, using both Hofmann and Curtius rearrangements on a carboxylic acidderivative?
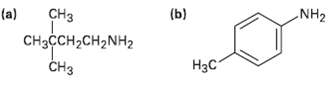
Transcribed Image Text:
CHз (b) (a) NH2 CняҫсH2CH2NH2 CHз Нас
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 41% (17 reviews)
In both of these reactions the product amine is for...View the full answer

Answered By

Stephen ouma
I have worked with different academic writing companies such as wriredom, writerbay, and Upwork. While working with these companies, I have helped thousands of students achieve their academic dreams. This is what I also intend to do here in SolutionInn
4.90+
19+ Reviews
63+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
How would you prepare the following carboxylic acids? (a) (CH 3 ) 3 CCO 2 H from (CH 3 ) 3 CCl (b) CH 3 CH 2 CH 2 CO2H from CH 3 CH 2 CH 2 Br
-
Using any alkyne needed, how would you prepare the following alkenes? (a) Trans-2-Octenc (b) Cis-3-Heptcne (c) 3-Methyl-1-pentene
-
How would you prepare the following diols? (b) (a)
-
The receiver most commonly used in AM and FM radio broadcast is the so-called superheterodyne receiver. Answer the following questions about this receiver. a. Draw the block diagram of a...
-
To identify instances of sexual harassment, the courts may use a "reasonable woman" standard of what constitutes offensive behavior. This standard is based on the idea that women and men have...
-
(a) A compound U (C9H10O) gives a negative iodoform test. The IR spectrum of U shows a strong absorption peak at 1690 cm-1. The 1H NMR spectrum of U gives the following: Triplet...
-
The risks of incorrect rejection and incorrect acceptance are related but involve two entirely different outcomes: Incorrect rejection means the risk of concluding that recorded book value is...
-
Beta Graphics, Inc., has the following data: Requirement 1. Perform a vertical analysis of Betas balance sheet for eachyear. BETA GRAPHICS, INC. Comparative Balance Sheet December 31, 2012 and 2011...
-
A 25 kg child slides, from rest, down a playground slide that is 4.0 m long, as shown in the figure. The slide makes a 40 angle with the horizontal. The child's speed at the bottom is 3.2 m/s. What...
-
The 'estimating trumpet' illustrates tolerances and expectations around levels of estimating accuracy across the life cycle. How do the tolerances resonate with your organisation's expectations...
-
How could you prepare the following amine using a reductive aminationreaction?
-
What products would you expect from Hofmann elimination of the following amines? If more than one product is formed, indicate which ismajor. NH2 (b) NH2 (a) CH3CH2CH2CHCH2CH2CH2CH3 NHCH2CH3 (d) NH2...
-
During 2014, Crest Corporation earned $ 5,000 of cash revenue and accrued $ 3,000 of salaries expense. Required Based on this information alone: a. Prepare the December 31, 2014, balance sheet. b....
-
A sample of blood is placed in a centrifuge of radius .15 m. The mass of a red corpuscle is 3.0 x 10 -16 kg, and the magnitude of the force required to make it settle out of the plasma is 4.0 x 10...
-
The first harmonic of a series created by a standing air wave in a tube open at one end only is 35.4 Hz. If the length of the tube is 218 cm, calculate the speed of sound of the air. Use two digits...
-
What angular velocity would result in a centripetal acceleration of 25 ft/s^2 if the radius of the curve is 100' ? What velocity in MPH is would be required to make a 33 degree elevated banked turn...
-
A black hole of ten solar masses is emitting blackbody radiation while also absorbing mass from a surrounding accretion disk. In order for the black hole to remain at a constant mass, what must be...
-
1. Suppose you measure three independent variables as x = 10 2, y = 7 1, = 40 3, and use these values to compute = + 2/ + sin (4) What should be your answer for and its uncertainty? 2. The Type K...
-
Which of the following is an acceptable method to report total comprehensive income according to U.S. GAAP? (a) On the face of the balance sheet. (b) In the discontinued operations section of the...
-
Design an experiment to demonstrate that RNA transcripts are synthesized in the nucleus of eukaryotes and are subsequently transported to the cytoplasm.
-
A sample of CaCO 3 (s) is introduced into a sealed container of volume 0.654 L and heated to 1000 K until equilibrium is reached. The K p for the reaction CaCO 3 (s) CaO(s)+ CO 2 ( g) is 3.9 * 10 -2...
-
The ammonium ion, NH4+, has a tetrahedral geometry analogous to that of methane. Explain this structure in terms of atomic and molecular orbitals.
-
Use lines, dashed wedges, and solid wedges to show the geometry of CF4 and CH3SH.
-
Silicon is just below carbon in the periodic table. Predict the geometry of silicon tetrafluoride, SiF4.
-
You are the new controller for Moonlight Bay Resorts. The company CFO has asked you to determine the company's interest expense for the year ended December 31, 2021. Your accounting group provided...
-
How do the dynamics change in Calm Down by Selena Gomez ?
-
A company's inventory records report the following: August 1 August 5 August 12 Beginning balance Purchase Purchase 23 units @ $13 18 units $12 22 units @ $13 On August 15, it sold 46 units. Using...

Study smarter with the SolutionInn App


