How would you prepare the following carbonyl compounds starting from an alkyne (reddish brown ? Br)? (b)
Question:
How would you prepare the following carbonyl compounds starting from an alkyne (reddish brown ? Br)?
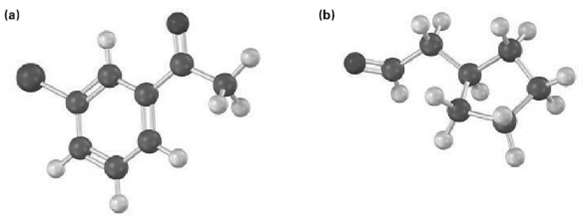
Transcribed Image Text:
(b) (a)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 44% (18 reviews)
a Br b HC ...View the full answer

Answered By

Asd fgh
sadasmdna,smdna,smdna,msdn,masdn,masnd,masnd,m asd.as,dmas,dma.,sd as.dmas.,dma.,s ma.,sdm.,as mda.,smd.,asmd.,asmd.,asmd.,asm
5.00+
1+ Reviews
15+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
How would you prepare the following carbonyl compounds from anitrile? ( (a) CH3CH2CCH2CH3 CH O2N
-
How would you prepare the following compounds from the given starting materials? a. b. c. d. CH3CH2CH CH3CHCH N(CH3)2 CH CHCH OCH3
-
Using any alkyne needed, how would you prepare the following alkenes? (a) Trans-2-Octenc (b) Cis-3-Heptcne (c) 3-Methyl-1-pentene
-
Assume that Polaris manufactures and sells 60,000 units of a product at $ 11,000 per unit in domestic markets. It costs $ 6,000 per unit to manufacture ($ 4,000 variable cost per unit, $ 2,000 fixed...
-
An architectural design firm is faced with a decision as to which projects to bid on for the coming year. Ten projects are available for which they are qualified. The following table lists the profit...
-
A cylindrical tank with hemispherical heads is constructed of steel sections that are welded circumferentially (see figure). The tank diameter is 1.25 m, the wall thickness is 22 mm, and the internal...
-
For what purposes do businesses use blogs?
-
For each of the unrelated transactions described below, present the entry(ies) required to record each transaction. 1. Grand Corp. issued $20,000,000 par value 10% convertible bonds at 99. If the...
-
What are some of of the Trends and Implications of Human Resources that are impacting the future ?
-
The three companies shown below have gathered data to determine whether they should factor their accounts receivable. Required: Prepare calculations for each company to determine the total cost of...
-
What alkyne would you start with to prepare each of the following compounds by a hydroboration/oxidationreaction? (b) (a) CH CH-CCHCH3 -CH2CH CH
-
The pK a of acetone, CH 3 COCH 3 , is 19.3. Which of the following bases is strong enough to de-protonate acetone? (a) KOH (p K a of H 2 O = 15.7) (b) Na + C CH (p K a of C 2 H 2 = 25) (c) NaHCO 3...
-
Prove that the duration of a portfolio is the weighted average duration of the portfolio assets.
-
Tau issued a statement to one of its customers, Ancona Co for the month ended 28 February 20X5. At that date, the payable ledger account for Tau maintained by Ancona Co had a balance of $4,575. A...
-
Suppose a car runs over a nail while driving at a speed of 6 2 miles per hour, and the nail is lodged in the tire tread 1 2 inches from the center of the wheel. What is the angular velocity of the...
-
A physics students with a mass of 7.1kg runs with a speed of 7.5kmh^-1 and jumps on a waveboard of mass 14.4kg sitting stationary at the edge of the pool, ignoring friction, what is the speed ofbthe...
-
9. Tallula is skydiving. The mass of Tallula and her parachute is 70.0 kg. She is falling at a constant speed of 6.30m. If she falls at this speed for 20.0 s and for a distance of 126 m, what is the...
-
6 . Sara goes to her apartment on the fifth floor by elevator from the ground floor. The whole journey is 1 8 . 0 meters. If Sara has a mass of 6 0 kg , what is the gravitational potential energy...
-
Picture a long, straight corridor running east-west, with a water fountain located somewhere along it. Starting from the west end of the corridor, a woman walks a short distance east along the...
-
The executor of Gina Purcells estate has recorded the following information: Assets discovered at death (at fair value): Cash . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . ....
-
Write balanced complete ionic and net ionic equations for each acidbase reaction. a. HBr(aq) + NaOH(aq) b. HF(aq) + NaOH(aq) C. HCHO(aq) + RbOH(aq)
-
Classify the following peptides as acidic, basic, or neutral. What is the net charge on each peptide at pH = 6? (a) Gly-Leu-Val (b) Leu-Trp-Lys-Gly-Lys (c) N-acetyl-Asp-Val-Ser-Arg-Arg (A-acetyl...
-
Classify each of the following pericyclic reactions as an electrocyclic, cycloaddition, or sig- matropic reaction. Give the curved-arrow notation for each reaction, and tell how many electrons are...
-
Show by a frontier orbital analysis that the [Aa + 2s] and [4s + 2a] modes of cycloaddition are not allowed.
-
What is the balance in Work in Process Inventory at the end of each month? Work in Process Inventory April 30 $ May 31 A 10,140 15,950 June 30 $ 10,450
-
Sako Company s Audio Division produces a speaker used by manufacturers of various audio products. Sales and cost data on the speaker follow: Selling price per unit on the intermediate market $ 6 0...
-
Vulcan Company's contribution format income statement for June is as follows: Vulcan Company Income Statement For the Month Ended June 30 Sales Variable expenses Contribution margin Fixed expenses...

Study smarter with the SolutionInn App


