Write balanced complete ionic and net ionic equations for each acidbase reaction. a. HBr(aq) + NaOH(aq) b.
Question:
Write balanced complete ionic and net ionic equations for each acid–base reaction.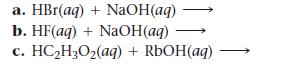
Transcribed Image Text:
a. HBr(aq) + NaOH(aq) b. HF(aq) + NaOH(aq) C. HC₂H₂O₂(aq) + RbOH(aq)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (4 reviews)
a Complete ionic Haq Br aq Nat aq OHaq HOl Na aq Bra...View the full answer

Answered By

Shameen Tahir
The following are details of my Areas of Effectiveness. The following are details of my Areas of Effectiveness English Language Proficiency, Organization Behavior , consumer Behavior and Marketing, Communication, Applied Statistics, Research Methods , Cognitive & Affective Processes, Cognitive & Affective Processes, Data Analysis in Research, Human Resources Management ,Research Project,
Social Psychology, Personality Psychology, Introduction to Applied Areas of Psychology,
Behavioral Neurosdence , Historical and Contemporary Issues in Psychology, Measurement in Psychology, experimental Psychology,
Business Ethics Business Ethics An introduction to business studies Organization & Management Legal Environment of Business Information Systems in Organizations Operations Management Global Business Policies Industrial Organization Business Strategy Information Management and Technology Company Structure and Organizational Management Accounting & Auditing Financial Accounting Managerial Accounting Accounting for strategy implementation Financial accounting Introduction to bookkeeping and accounting Marketing Marketing Management Professional Development Strategies Business Communications Business planning Commerce & Technology Human resource management General Management Conflict management Leadership Organizational Leadership Supply Chain Management Law Corporate Strategy Creative Writing Analytical Reading & Writing Other Expertise Risk Management Entrepreneurship Management science Organizational behavior Project management Financial Analysis, Research & Companies Valuation And any kind of Excel Queries.
4.70+
16+ Reviews
34+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Write complete ionic and net ionic equations for each of the following molecular equations. a. 2HNO3(aq) + Mg(OH)2(s) 2H2O(l) + Mg(NO3)2(aq) Nitric acid, HNO3, is a strong electrolyte. b....
-
Write balanced complete ionic and net ionic equations for each reaction. a. HCl(aq) + LiOH(aq) HO(1)+ LiCI(aq) b. Cas(aq) + CuCl(aq) CuS(s) + CaCl(aq) c. NaOH(aq) + HCHO(aq) d. Na3PO4(aq) + NiCl(aq)...
-
Write balanced complete ionic and net ionic equations for each acidbase reaction. a. HI(aq) + RbOH(aq) b. HCHO(aq) + NaOH(aq) c. HCHO(aq) + LiOH(aq)
-
1. Using a diagram of utility maximization, show: a. The income effect of price change b. The substitution effect of price change
-
Discuss what you consider to be the major differences between preventive, detective, and corrective control procedures. Give two examples of each type of control.
-
How are placer gold deposits formed?
-
Distinguish between general and application controls and indicate the relative importance of each category.
-
Eleby Company has gathered the following information. Units in beginning work in process .. 0 Units started into production ... 36,000 Units in ending work in process .... 6,000 Percent complete for...
-
Give a simple description of the language generated by the following context-free grammar: S aSb|bSa|SS| and prove that it does in fact generate that language. Once you know the language, the...
-
A 25.00-mL sample of an unknown HClO 4 solution requires titration with 22.62 mL of 0.2000 M NaOH to reach the equivalence point. What is the concentration of the unknown HClO 4 solution? The...
-
Complete and balance each acidbase equation. a. HI(aq) + LiOH(aq) b. HCHO(aq) + Ca(OH)(aq) c. HCl (aq) + Ba(OH)(aq)
-
AT&T Inc. (T) is a leading global provider of telecommunication services. Facebook, Inc. (FB) is a major worldwide social media company. AT&T has a lengthy history and was founded by Alexander Graham...
-
For the following exercise, use the logistic growth model f(x) 14. Find and interpret f(4). Round to the nearest tenth. = 150 -2x 1+8e
-
5. If you are multiplying a matrix A by a vector, what is the relationship between the the number of columns in A and the dimension of ?
-
Determine if V 1 = ( 2 , - 1 , 3 ) V 2 = ( 4 , 1 , 2 ) V 3 ( 8 , - 1 , 8 ) Form a set that spans R ^ 3
-
For the matrix 1 1 1 1 X' = 4 -2 3-5 compute P= X(X'X)-'X' and M = I-P (a) Verify that MP = 0. (b) Let Q = 1 3 2 8 and X = XQ. Compute the P and M based on X instead of X.
-
Assume f(x) is the closed-form expression for the following summation n 3. f(x) = x=1+ x + x + x + ... + x " i=0 And let g(x) represent the closed-form expression for the following summation: n 9(x)...
-
Why would an auditor gather evidence using a combination of internal control testing and substantive testing?
-
Explain the operation of the dividends received deduction.
-
Calculate the kinetic energy in Nm of a 75-kg box moving on a conveyor at 6.85 m/s. The formula for kinetic energy is KE = mv 2 , where m = mass and v = velocity.
-
Calculate the mass of a body in kg if it has a kinetic energy of 38.6 Nm when moving at 31.5 km/h. The formula for kinetic energy is KE = mv 2 , where m = mass and v = velocity.
-
Calculate the mass of a body in g if it has a kinetic energy of 94.6 m Nm when moving at 2.25 m/s. The formula for kinetic energy is KE = mv 2 , where m = mass and v = velocity.
-
The function depicted below searches for a given value x in the sorted array arr - What is the worst-case time complexity of the function? (4 marks) int function(int arr[], int 1, int r, int x) { 1 2...
-
How do feminist sociological perspectives critique traditional gender norms, and what are the key challenges in achieving gender equity within deeply patriarchal structures that permeate cultural,...
-
1. (5pt) Find the Fourier series of $f$ on the given interval. $$ f(x)=\left\{\begin{array}{c} 1-x, & -1

Study smarter with the SolutionInn App


