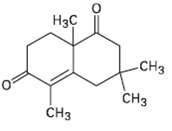
How would you prepare the following compound using a Robinson annulations reaction between a ?-dike tone and
Question:
How would you prepare the following compound using a Robinson annulations reaction between a ?-dike tone and ?, ?-unsaturated ketone? Draw the structures of both reactants and the intermediate Michael addition product.
Transcribed Image Text:
Нас .CHз CHз CHз
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 88% (18 reviews)
CH3CHCCHCH Michael acceptor H2O HC H Michael donor H3C CH3 CH3 C...View the full answer

Answered By

Poonam Chaudhary
I have 15 month+ Teaching Experience
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
How would you prepare the following compound using a Michaelreaction?
-
How would you prepare the following compound using an acetoacetic estersynthesis?
-
Using any alkyne needed, how would you prepare the following alkenes? (a) Trans-2-Octenc (b) Cis-3-Heptcne (c) 3-Methyl-1-pentene
-
Brandon Stroud was driving a golf car made by Textron, Inc., to transport guests at Christmas party. The golf car did not have lights, but Textron did not warn against using it on public roads at...
-
Matrix organizations first sprang up in businesses that worked on scientific and engineering projects for narrow customer groups. Examples include Fluor, which built oil refineries in Saudi Arabia,...
-
Consider the circuit in Fig. 4.145. (a) Replace the resistor RL by a zero resistance ammeter and determine the ammeter reading. (b) To verify the reciprocity theorem, interchange the ammeter and the...
-
Consider the conditions of Problem 7.36, but now allow for radiation exchange between the surface of the heating element \((\varepsilon=0.8)\) and the walls of the duct, which form a large enclosure...
-
You decide to help I. M. with his analysis. A good fax machine will cost $500 and functions properly for five years. The phone company charges $300 for installing a new line and $60 a month for the...
-
Use the May 31 fiscal year-end information from the following ledger accounts (assume that all accounts have normal balances). General Ledger Retained Earnings Date May 31 PR Debit Account Number 318...
-
The demand functions for two commodities, A and B, are given by QA = AP 0.5 Y 0.5 and Q B = BP 1.5 Y 1.5 where A and B are positive constants (a) Find the price elasticity of demand for each good and...
-
What product would you expect from a Robinson annulations reaction of 2-methyl-1, 3-cyclopentanedione with3-buten-2-one? -CH + H%3DH 2-Methyl-1,3-cyclo- pentanedione 3-Buten-2-one
-
What ketone or aldehydes might the following enone have been prepared from by aldolreaction? (b) (a)
-
At the end of 2018 the following information is available for Billings and Phoneix companies: Required a. Prepare common size income statements for each company. b. One company is a high-end...
-
What type of audit is used by the insurance company after the claims have been paid?
-
(Yield to maturity) You own a 22-year bond that pays 9 percent interest annually. The par value of the bond is $1,000 and the market price of the bond is $1,000. What is the yield to maturity of the...
-
What industry led the push for the incorporation of as little iron as possible in building construction in the interest of keeping costs down?
-
A flat fee associated with a specific, routine event, like $25 for a doctor's visit. The maximum amount of medical expenses you can pay each year. The percentage of your healthcare costs you cover....
-
Why are gains/losses added back to (or deducted from) the cash from operations section of a cash flow statement?
-
What do you think Lululemon might need to do better to expand rapidly in the global marketplace?
-
On March 31, 2018, Gardner Corporation received authorization to issue $30,000 of 9 percent, 30-year bonds payable. The bonds pay interest on March 31 and September 30. The entire issue was dated...
-
List these compounds in order of increasing carboncarbon bond strength and in order of decreasing carboncarbon bond length: HCCH, H 2 CCH 2 , H 3 CCH 3 .
-
Allylic halides have the structure (a) Show how the first-order ionization of an allylic halide leads to a resonance-stabilized cation. (b) Draw the resonance structures of the allylic cations formed...
-
List the following carbocations in decreasing order of their stability. CH +CH, +CH, CH CH
-
Two of the carbocations in Problem 6-53 are prone to rearrangement. Show how they might rearrange to more stable carbocations.
-
x-8x+15 A) Let f(x) = = x2+2x-15 Calculate lim f(x) x 3 2x225x75 B) Let f(x) = x-18x45 Calculate lim f(x) x 15
-
If a dance instructor prices her lessons at $60 per student, she will have three students. If she prices her lessons at $50 per student, she will have four students. How much marginal revenue will...
-
What is the difference between objectives, strategies, and goals? How do these things improve our planning process?

Study smarter with the SolutionInn App


