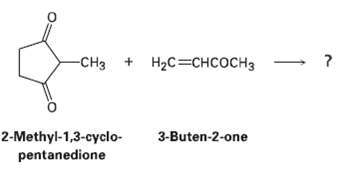
What product would you expect from a Robinson annulations reaction of 2-methyl-1, 3-cyclopentanedione with3-buten-2-one? -CH + H%3DH
Question:
What product would you expect from a Robinson annulations reaction of 2-methyl-1, 3-cyclopentanedione with3-buten-2-one?
Transcribed Image Text:
-CHз + HаС%3DснсосHз 2-Methyl-1,3-cyclo- pentanedione 3-Buten-2-one
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 73% (15 reviews)
The Robinson annulation is a combination of two reactions covered in this chapter F...View the full answer

Answered By

Somshukla Chakraborty
I have a teaching experience of more than 4 years by now in diverse subjects like History,Geography,Political Science,Sociology,Business Enterprise,Economics,Environmental Management etc.I teach students from classes 9-12 and undergraduate students.I boards I handle are IB,IGCSE, state boards,ICSE, CBSE.I am passionate about teaching.Full satisfaction of the students is my main goal.
I have completed my graduation and master's in history from Jadavpur University Kolkata,India in 2012 and I have completed my B.Ed from the same University in 2013. I have taught in a reputed school of Kolkata (subjects-History,Geography,Civics,Political Science) from 2014-2016.I worked as a guest lecturer of history in a college of Kolkata for 2 years teaching students of 1st ,2nd and 3rd year. I taught Ancient and Modern Indian history there.I have taught in another school in Mohali,Punjab teaching students from classes 9-12.Presently I am working as an online tutor with concept tutors,Bangalore,India(Carve Niche Pvt.Ltd.) for the last 1year and also have been appointed as an online history tutor by Course Hero(California,U.S) and Vidyalai.com(Chennai,India).
4.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
What product would you expect from reaction of cyclohexene with HBr? With HC1 HBr
-
What product would you expect from the reaction of butyrolactone with LiAlH4 with DIBAH? Butyrolactone
-
What product would you expect from the reaction of cyclopentane with NBS and water? Show the stereo chemistry.
-
Consider the 70 households that purchased a DVD player, 48 households are satisfied with their purchase and 22 households are dissatisfied. Suppose two households are randomly selected from the...
-
It is sometimes argued that a matrix organization can serve as a mechanism for achieving strategic fit - the achievement of synergies across related business units resulting in a combined performance...
-
It has been suggested that the liquid flow rate in a large diameter pipeline could be better regulated by using two control valves instead of one. Suppose that one control valve has a large C v...
-
Describe whys the medical record is important in legal proceedings.
-
You are a new summer intern working for a major professional services firm. During your lunch break each day, you and a fellow intern, Bob, eat at a local sandwich shop. One day, Bobs girlfriend...
-
Define and discuss the terms 'privacy' in the context of web analytics by drawing on an example.
-
The chapter discussed a study that compared and contrasted people who lead happy lives versus those with meaningful lives. How does this distinction relate to the way you decide to spend your time...
-
Show how you might use an enamine reaction to prepare each of the followingcompounds: (b) (a) CH2CH2CO2CH3 CH2CH2CN
-
How would you prepare the following compound using a Robinson annulations reaction between a ?-dike tone and ?, ?-unsaturated ketone? Draw the structures of both reactants and the intermediate...
-
Distinguish among the shapes of a normal yield curve, an abnormal curve, and a humped curve.
-
The temperature (in oC) in a certain city follows a Normal distribution with mean 20. Which would be the standard deviation if we know that the probability that the temperature is between 16 and 24...
-
Equivalent Taxable Yield rm = r(1-t) r = rm/(1-t) Example: Suppose your tax rate is 25% on a taxable corporate bond. A municipal bond is available paying 4% interest. What interest rate would you...
-
10. A security generates a fixed yet unknown cash flow of some amount for each of the first three years (i.e. t-1, t-2, and t-3), and then $1,000 per year for each of the remaining seven years (t-4...
-
How much money is needed to establish a fund paying $80,000 annually forever with the first payment made immediately if the fund will earn interest at 5% p.a. compounded semi-annually?
-
Assume you can choose one of the following payment alternatives for a project that Receiving $4,000 five years from now (Year 5) Receiving $800 each year at the end of Year 1, 2, 3, and 4. Receiving...
-
Jorge is a resident and citizen of Spain. He invests $500,000 in Miami Beach real estate. When Jorge died in 2017, he owned $6,000,000 in assets in Spain in addition to the Miami Beach real estate.
-
On April 29, 2015, Auk Corporation acquires 100% of the outstanding stock of Amazon Corporation (E & P of $750,000) for $1.2 million. Amazon has assets with a fair market value of $1.4 million (basis...
-
Hydrogenation reactions are used to add hydrogen across double bonds in hydrocarbons and other organic compounds. Use average bond energies to calculate H rxn for the hydrogenation reaction. HC =...
-
For each pair of compounds, predict which one has the higher molecular dipole moment, and explain your reasoning. (a) Ethyl chloride or ethyl iodide (b) 1-bromopropane or cyclopropane (c) Cis-2,...
-
Chlorocyclohexane reacts with sodium cyanide (NaCN) in ethanol to give cyanocyclohexane. The rate of formation of cyanocyclohexane increases when a small amount of sodium iodide is added to the...
-
Give the solvolysis products expected when each compound is heated in ethanol. (a) (b) (c) (d) Br Cl CH
-
Suppose the North Carolina University system tests the idea that computerization of statistics classes increases student performance in the courses. If the computers are found to have a beneficial...
-
M. Handy is a self-employed painter who earned $32,400 last year. Her FICA tax rate is 15.3% of her earnings. How much FICA tax did she pay?
-
Using the same iThe Shouldice Hospital specializes in hernia operations. They have a special procedure that not only has a much higher rate of long-term success than other hospitals, but also allows...

Study smarter with the SolutionInn App


