How would you prepare the following compounds fromCyclohexanone? la) (b) o C6H5CH, CHC&H5 CH2CH2CN (c) (d) CH2CH=CH2
Question:
How would you prepare the following compounds fromCyclohexanone?
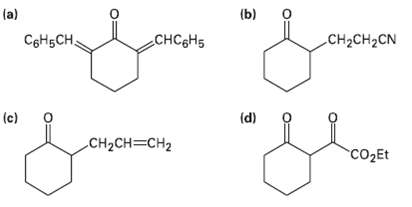
Transcribed Image Text:
la) (b) o C6H5CH, CHC&H5 CH2CH2CN (c) (d) CH2CH=CH2 CO2Et
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (7 reviews)
So 1 NaOH CsH5CH EtOH 2 CoHsCHO ...View the full answer

Answered By

Fahmin Arakkal
Tutoring and Contributing expert question and answers to teachers and students.
Primarily oversees the Heat and Mass Transfer contents presented on websites and blogs.
Responsible for Creating, Editing, Updating all contents related Chemical Engineering in
latex language
4.40+
8+ Reviews
22+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
How would you prepare the following compounds starting with an appropriate carboxylic acid and any other reagents needed? (Reddish brown =Br.) (a) (b)
-
How would you prepare the following compounds using either an acetoacetic ester synthesis or a malonic estersynthesis? (d) (c) ( (a) H CHO2Et H H2H,H3 "CH Et
-
How would you prepare the following compounds from toluene? A diazonio replacement reaction is needed in someinstances. (a) (b) (c) NH2 CH2NH2 H
-
The emergency room of the community hospital in Farmburg has one receptionist, one doctor, and one nurse. The emergency room opens at time zero, and patients begin to arrive some time later. Patients...
-
Coke and Pepsi have sustained their market dominance for nearly a century. General Motors and Ford have recently been hard hit by competition. What is different about the product/market situation in...
-
The following electron configurations represent excited states. Identify the element, and write its ground-state condensed electron configuration. (a) 1s2 2s2 3p2 4p1, (b) [Ar] 3d10 4s1 4p4 5s1, (c)...
-
What are the four dimensions of ITIL4?
-
Luce & Morgan, a law firm in downtown Jefferson City, is considering opening a legal clinic for middle-and low-income clients. The clinic would bill at a rate of $18 per hour. It would employ law...
-
On December 31, Jarden Company's Allowance for Doubtful Accounts has an unadjusted credit balance of $15,500. Jarden prepares a schedule of its December 31 accounts receivable by age. Accounts...
-
You, CPA, work as a consultant on various engagements. Your client, Over The Edge Ltd. (OTE), has grown from a small custom snowboard manufacturer servicing the local market to a multinational...
-
Fill in the missing reagents a?h in the following scheme: CO2Et CH3 CO2Et 2. f 1. c 2. d 1. a 2. b 1. g 1. e 2. h CO2Et
-
Leucine, one of the twenty amino acids found in proteins, is metabolized by a pathway that includes the following step. Propose amechanism. "02C "C H3C SCOA SCOA 3-Hydroxy-3-methyl- glutaryl CoA...
-
Consider the precedence graph of Figure is the corresponding schedule conflict serializable? Explain your answer. T2 T1 T4 T5
-
Dora is hired by Nancy to work in Nancy's pizza delivery operation. Nancy does a thorough background check on Dora and learns that Dora has a perfect driving record with no complaints from any of her...
-
Able, Baker, and Charlie each operate their own landscaping businesses as sole proprietors. The three of them were each hired by one client for doing a particularly big job. The three of them are...
-
Locate Victoria State's Code of conduct of conveyancing and download a copy. Explain which state and what the title of the Code of conduct is and where it is found. When is its operational start...
-
Question 1. Ali ahmad and chong have decided to form a business for selling books at putrajaya. they confused on which types of business entities are more suitable for their businesses. advise ali,...
-
Drafting a Simple Business Contract Background You are the business owner. You offer sitter services for elderly. Your client has 85 yr old healthy dad who needs a companion from 1-5 pm, 5 days a...
-
What is the rotational inertia about the \(x\) axis of the rigid object in Figure P11.53? (Treat the balls as particles.) Data from Figure P11.53 Figure P11.53 m 54m
-
Information graphics, also called infographics, are wildly popular, especially in online environments. Why do you think infographics continue to receive so much attention? How could infographics be...
-
Draw the Lewis structure for CO with an arrow representing the dipole moment. Refer to Figure 10.10 to estimate the percent ionic character of the CO bond. Percent ionic character 100 75 50 25 0. HI...
-
Give the systematic (IUPAC) names of the following alkenes. (a) (b) (c) (d) (e) (f) (g) (h) (i) (j) CH2 CHCH2CH(CH3)2 CH3(CH2)3-C-CH2CH3 CH2 CH CHCH2 CH CH2 CH2=C=CH-CH-CH2 " CH3 CH3
-
A chemist allows some pure (2S,3R)-3-bromo-2,3-diphenylpentane to react with a solution of sodium ethoxide (NaOCH2CH3) in ethanol. The products are two alkenes: A (cis-trans mixture) and B, a single...
-
The energy difference between cis- and trans-but-2-ene is about 4 kJ mol; however, the trans isomer of 4,4-dimethylpent-2-ene is nearly 16 kJ mol more stable than the cis isomer. Explain this large...
-
A skier glides down a frictionless hill of 100 meters high, then ascends another hill, with a height of 90 meters, as shown in the figure below. a) What is the speed of the skier when they reach the...
-
The atoms in a nickel crystal vibrate as harmonic oscillators with an angular frequency of 5.5x 1013 rad/s. The mass of a nickel atom is 9.75 x 10-26 kg. What is the difference in energy between...
-
Ballistic pendula are used by forensic analysts to calculate the velocity of a bullet. The bullet is shot into a target suspended by a long, thin, narrow rod; the degree to which the pendulum is...

Study smarter with the SolutionInn App


