Draw the Lewis structure for CO with an arrow representing the dipole moment. Refer to Figure 10.10
Question:
Draw the Lewis structure for CO with an arrow representing the dipole moment. Refer to Figure 10.10 to estimate the percent ionic character of the CO bond.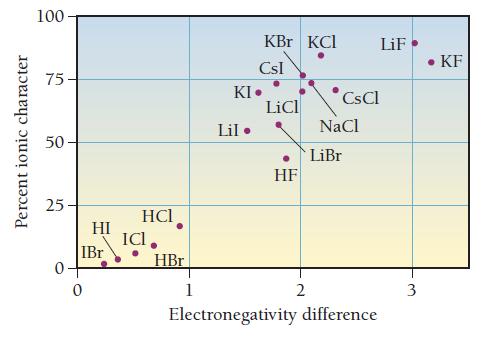
Transcribed Image Text:
Percent ionic character 100 75 50 25 0. HI IBr 0 HCI ICI. HBr 1 KBr KCI CsI KI. Lil. LiCl HF 2 CsCl NaCl LiBr LiF Electronegativity difference 3 • KF
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)

Answered By

Muhammad Haroon
More than 3 years experience in teaching undergraduate and graduate level courses which includes Object Oriented Programming, Data Structures, Algorithms, Database Systems, Theory of Automata, Theory of Computation, Database Administration, Web Technologies etc.
5.00+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Draw the Lewis structure for BrF with an arrow representing the dipole moment. Refer to Figure 10.10 to estimate the percent ionic character of the BrF bond. Percent ionic character 100 75 50 25 0....
-
The ionic character of the bond in a diatomic molecule can be estimated by the formula Where m is the experimentally measured dipole moment (in C m), e the electronic charge, and d the bond length in...
-
The Lewis structure for allene is Make a sketch of the structure of this molecule that is analogous to Figure 9.25. In addition, answer the following three questions: (a) Is the molecule planar? (b)...
-
Problem 1: The three velocities shown would indicate that the body is not rigid. Given that the body is known to be rigid, one of the three velocities is clearly incorrect. Draw all lines and take...
-
Souder, Oles, and Franek is an international consulting firm headquartered in Chicago, Illinois. The Entity-Relationship diagram in Figure shows a simplified version of the companys process for...
-
Refer to the financial statements and other data in Problem 1612. Assume that you are an account executive for a large brokerage house and that one of your clients has asked for a recommendation...
-
Johanna Marra and Eric Nazzaro began a romantic relationship in October 2013. That previous July, Nazzarro had purchased a duplex that he intended to renovate. Nazzarro rented out the top floor while...
-
Lifemaster produces two types of exercise treadmills: regular and deluxe. The exercise craze is such that Lifemaster could use all its available machine hours to produce either model. The two models...
-
- Suppose that Brine Freez servings sell for $4 each; capital is paid a daily rental rate of $20 ; and labor is paid a daily wage of $80 . - Calculate the value of marginal product of labor (L) and...
-
Determine if a bond between each pair of atoms would be pure covalent, polar covalent, or ionic. a. C and N b. N and S c. K and F d. N and N
-
Determine if a bond between each pair of atoms would be pure covalent, polar covalent, or ionic. a. Br and Br b. C and Cl c. C and S d. Sr and O
-
A spring has k = 88 N/m. Use a graph to determine the work needed to stretch it from x = 3.8cm to x = 5.8cm, where x is the displacement from its unstretched length.
-
Out of 10 pay telephones located in a municipal building, two phones are to be picked at random, with replacement, for a study of phone use. The actual usage of the phones on a particular day is...
-
A population has a variance 2 of 100. A sample of 25 from this population had a mean equal to 17. Can we reject H 0 : = 21 in favor of H 1 : 21? Let = .05.
-
A 1984 study indicated that the average yearly housing cost for a family of 4 was $12,983. A random sample of 200 families in a US city resulted in a mean of $ 14,039 with a standard deviation of...
-
Briefly explain what is meant by the power of a test. Why is the power of the test important?
-
Topgun Records and several movie studios have decided to sign a revenue-sharing contract for CDs. Each CD costs the studio $2 to produce. The CD will be sold to Topgun for $3. Topgun, in turn, prices...
-
Define demand-side and supply-side policies. Sometimes it is said that Keynesians advocate demand-side policies and monetarists advocate supply-side policies. Is there any accuracy in this statement?
-
For the vector whose polar components are (Vr = 1, Vθ = 0), compute in polars all components of the second covariant derivative Vα;μ;ν. To find...
-
A pressure relief valve is designed so that the gas pressure in the tank acts on a piston with a diameter of 30 mm. How much spring force must be applied to the outside of the piston to hold the...
-
A gas-powered cannon shoots projectiles by introducing nitrogen gas at 20.5 MPa into a cylinder having an inside diameter of 50 mm. Compute the force exerted on the projectile.
-
The egress hatch of a manned spacecraft is designed so that the internal pressure in the cabin applies a force to help maintain the seal. If the internal pressure is 34.4 kPa(abs) and the external...
-
Suppose 1 an converges. Prove that lim r-1, r <1 n=1 ran = An. n=1
-
Problem Solving An MP3 player Cost R2699. How much will R796 MP3 player cost? A charity organisation Collects 628 boxes of gifts. The content of each box is worth R5250. What is the Value of all the...
-
Let (an) be the sequence from the previous question. To show by induction that - an = 2 + 3 for all natural n, the induction assumption is ak = = 2k +3 and ak = 2ak-1 - 3 for some natural k > 1. ak...

Study smarter with the SolutionInn App


