Identify each of the following groups as an activator or deactivator and as and o,p-director orm-director. (e)
Question:
Identify each of the following groups as an activator or deactivator and as and o,p-director orm-director.
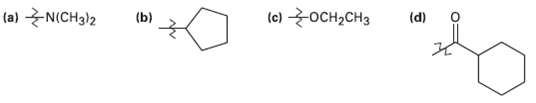
Transcribed Image Text:
(e) OCH2CH3 (a) N(CH3)2 (b) (d)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 90% (10 reviews)
Group a NCH32 b c CHCH3 d 0 Identification opactivator opactivator opactivator mdeactivat...View the full answer

Answered By

Madhvendra Pandey
Hi! I am Madhvendra, and I am your new friend ready to help you in the field of business, accounting, and finance. I am a College graduate in B.Com, and currently pursuing a Chartered Accountancy course (i.e equivalent to CPA in the USA). I have around 3 years of experience in the field of Financial Accounts, finance and, business studies, thereby looking forward to sharing those experiences in such a way that finds suitable solutions to your query.
Thus, please feel free to contact me regarding the same.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Identify each of the following groups as a population or a sample. If the group is a sample, identify the population to which the sample is related. A. The S&P MidCap 400 Index viewed as representing...
-
Identify each of the following as describing corporations, proprietorships, and/or partnerships. Some items have more than one answer. a. Distinct legal entity separate from its owners. b. More than...
-
Identify each of the following alkyl groups as being primary, secondary, or tertiary, and give it a systematic IUPAC name. (a) (b) (c) (d) (e) (f) CH CH3 CH3 CH CH2 CH2 CH3 CH3 CH3 CHCH CH3 CH2 CH3...
-
Use the data given in Table 3.9 to compute the mean, standard deviation, coefficient of variation, and coefficient of skewness for the current ratio of JNJ. Table 3.9 Year 1990 1991 1992 1993 1994...
-
William is employed by an accounting firm and uses his automobile in connection with his work. During the month of October 2016, he works at the office for 3 days and participates in the audit of a...
-
Again, like the previous problem, on February 4 of a particular year, the spot rate for U.S. dollars ($) expressed in euros () was $0.7873/. The U.S. interest rate (compounded annually) was 5.36...
-
a. Explain the auditor's responsibility for the quality and condition of the inventory. b. Indicate how the auditor can discover obsolete and slow moving items.
-
Keegan Corporations accounting records disclosed the following information for 2016: Cash sales ......................$850,000 Net credit sales .................... 720,000 Accounts receivable...
-
At the beginning of the year, the net assets of Shannon Company were $368,700. The only transactions affecting stockholders' equity during the year were net income of $42,000 and dividends of...
-
A tubular shaft similar to that shown in Figure 16.11 is to be designed that has an outside diameter of 80 mm (3.15 in.) and a length of 0.75 m (2.46 ft). The mechanical characteristic of prime...
-
The following compound can?t be synthesized using the methods discussed in this chapter. Why not?
-
Predict the major product(s) of nitration of the following substances. Which react faster than benzene and which slower? (a) Bromobenzene (b) Benzonitrile (c) Benzoic acid (d) Nitrobenzene (e)...
-
Sustainable Growth Based on the following information calculates the sustainable growth rate for Hendrix Guitars, Inc.: Profit margin = 6.4% Total asset turnover = 1.70 Total debt ratio = .40 Payout...
-
Raina borrowed $2000 from a lender that charged simple interest at an annual rate of 9%. When Raina paid off the loan, she paid $540 in interest. How long was the loan for, in years?
-
There are internal and external key factors that can impact negatively for a client's financial plan. Describe how you will make aware of these factors
-
The required rate of return of the market portfolio is 15% and the required rate of return of Beximco Pharmaceutical Limited (BPL) is 20%. Assuming that the risk free rate of return is 7%, find out...
-
If water begins to be valued as a basic necessity, would it be the same as if it were copper or oil or wheat? Explain
-
Explain why the rating on the water treatment plant bonds issued by Jackson Mississippi is lower than the rating on the general obligation bonds of the city. Comment on whether the two ratings will...
-
Select one of the preferred stocks listed in Table 16.1assume the dividends qualify for the preferential tax rate. Using the resources at your campus or public library or on the Internet, determine...
-
A Alkynes can be made by dehydrohalogenation of vinylic halides in a reaction that is essentially an E2 process. In studying the stereochemistry of this elimination, it was found that...
-
Redo Problem 5.29 using Aspen Plus. Problem 5.29 The inlet to an adiabatic compressor is nitrogen at 1 bar (0.1 MPa) and 150 K, and the exit pressure is 10 bar (1 MPa). a. Determine the minimum work...
-
Show the products of these reactions: Br DMSO + CH,CO, a) CI acetone + CH,CH,CO, b) CO DMF c) CH,CH CH,CH,Br +
-
Show all the steps in the mechanism for the reactions of 2-methyl-2-butanol with HCl in water.
-
Suggest reagents that could be used to prepare these alkyl halides from alcohols: CH3 CH, b) CH,CI ) C,r Ph CH;CH2 Br CI Br ) d)
-
Fields Company has two manufacturing departments, forming and painting. The company uses the weighted method and it reports the following unit data for the Forming department. Units completed in the...
-
Discuss and describe a specific process for creating artwork that involved procedures for combining diverse materials; describe in detail the selected product you posted. Part II Discuss and cite an...
-
What mechanisms do cells use to regulate vesicle trafficking within the endomembrane system, and how do SNARE proteins contribute to vesicle fusion ?

Study smarter with the SolutionInn App


