Show the products of these reactions: Br DMSO + CH,CO, a) CI acetone + CH,CH,CO, b)
Question:
Show the products of these reactions:
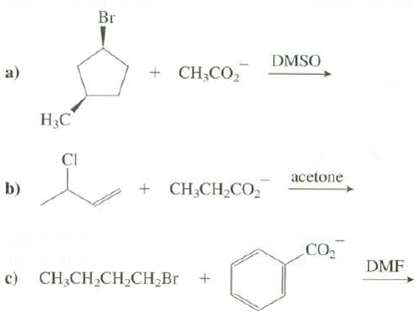
Transcribed Image Text:
Br DMSO + CH,CO, a) НС CI acetone + CH,CH,CO, b) CO DMF c) CH,CH CH,CH,Br +
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (8 reviews)
a H3C O...View the full answer

Answered By

Mario Alvarez
I teach Statistics and Probability for students of my university ( Univerisity Centroamerican Jose Simeon Canas) in my free time and when students ask for me, I prepare and teach students that are in courses of Statistics and Probability. Also I teach students of the University Francisco Gavidia and Universidad of El Salvador that need help in some topics about Statistics, Probability, Math, Calculus. I love teaching Statistics and Probability! Why me?
** I have experience in Statistics and Probability topics for middle school, high school and university.
** I always want to share my knowledge with my students and have a great relationship with them.
** I have experience working with students online.
** I am very patient with my students and highly committed with them
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Show the products of these reactions and explain whether each would follow an SN1 or an SN2 mechanism: a) C) B CI + OH Br + SH DMF CHOH HO Br + HO CHOH HO b) d) f) CI + HO CH,OH OTS + CH0 Br + CHCO...
-
Show the products of these reactions of 1-propylcyclopentene with thesereagents: a) Br, CCl, b) Br2, H2O d) HBr f) [OsO,], t-BuOOH c) 1) BH3, THF; 2) H,O,, NaOH e) H2O, H,SO, g) 1) O; 2) (CHz)S
-
Show the products of these reactions: CH,OH a) (CH,CH),NH + CH,CH,Br ether b) CH;CH,NCH3 + CH,I CH3
-
Stock W, X and Y have expected returns of 9.0%, 16.1% and 11.3% respectively. Based on this and the attached information, what is the expected return of your portfolio? Stock W X Y Number of Shares...
-
Explain the impact of a decrease in the level of activity on: (a) Total fixed cost. (b) Fixed cost per unit of activity.
-
In a random sample of eight people, the mean commute time to work was 35.5 minutes and the standard deviation was 7.2 minutes. You are given the sample mean and the sample standard deviation. Assume...
-
Discuss the essential steps for managing crises in projects.
-
Price, Inc., bottles and distributes mineral water from the companys natural springs in northern Oregon. Price markets two products: 12-ounce disposable plastic bottles and 1-gallon reusable plastic...
-
The budget process and budgets themselves can impact management actions, both positively and negatively. For instance, a common practice among businesses is for management to spend any amounts...
-
The following classification scheme typically is used in the preparation of a balance sheet: a. Current assets b. Investments and funds g. c. Property, plant, and equipment d. Intangible assets i....
-
Because of the acidic conditions, this reaction proceeds by an SN1 mechanism. Which hydroxy group acts as the leaving group in the reaction? Show all the steps in the mechanism for this reaction: ,,
-
Show all the steps in the mechanism for the reactions of 2-methyl-2-butanol with HCl in water.
-
A solution of 0.50 g of (-) - epinephrine (see Figure 5-15) dissolved in 10.0 mL of dilute aqueous HCl was placed in a 20-cm polarimeter tube. Using the sodium D line, the rotation was found to be...
-
An object moving in a liquid experiences a linear drag force: B = (bv, direction opposite the motion), where b is a constant called the drag coefficient. For a sphere of radius R, the drag constant...
-
In an amplified circuit, the current i (in A) changes with time t (in s) according to the equation below. If 0.085 C of charge has passed a point in the circuit at t=0, find the total charge to have...
-
Use the from step b) to compute the monthly excess outperformance (or underperformance in case the number is negative) of Fund X. The formula is: RX - RVG (as a reminder Rx and Rvg are excess returns...
-
Create an estimated retirement budget using a compound interest inflation rate of 3%. Estimate a retirement date based upon your projected retirement date 2060. Using the monthly spending...
-
Rick, a college financial aid representative, knows all the appropriate forms and procedures to help students apply for jobs on campus. What type of power does Rick hold for the students?
-
Air enters a combustion chamber at 75 m/s, 150 kPa and 300 K. Heat addition in the combustor amounts to 900 kJ/kg. Compute (a) the mass flow rate, (b) the exit properties and (c) amount of heat to be...
-
What is the purpose of the journal wizard?
-
20 British pounds is equal to $26.12. How many dollars is 12 pounds?
-
The double bond of an enamine (alkene amine) is much more nucleophilic than a typical alkene double bond. Assuming that the nitrogen atom in an enamine is sp2-hybridized, draw an orbital picture of...
-
Benzene has an ultraviolet absorption at ? max = 204mm, and para-toluidine has ? max = 235nm. How do you account for this difference? -NH2 p-Toluidine U max = 235 nm) Benzene Amax = 204 nm)
-
What product would you expect to obtain from a nucleophilic substitution reaction of (S)-2-bromohexane with acetate ion, CH3CO2 ? Assume that inversion of configuration occurs, and show the stereo...
-
What theorists are most likely to argue the city's form and growth come from decisions made by people and organizations that control wealth and other key resources?
-
Explain the difference between glucogenic and ketogenic amino acids
-
Describe what is an arrangement where workers who don't join a union must make payments equal to union dues and fees to get union representation services?

Study smarter with the SolutionInn App


