Identify the amino acid that is a catabolic precursor of each of the following ?-keto acids: (b)
Question:
Identify the amino acid that is a catabolic precursor of each of the following ?-keto acids:
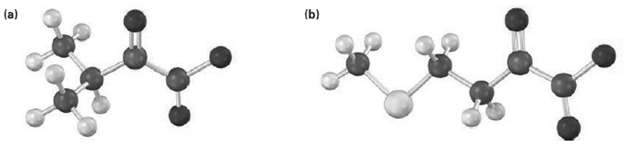
Transcribed Image Text:
(b) (a)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 36% (11 reviews)
The amino acid precursors are valine a and methionin...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
For each group of amino acids, identify the amino acid with the lowest pI (try to solve this problem by inspecting their structures, rather than performing calculations). (a) Alanine, aspartic acid,...
-
Draw the form of the amino acid that is expected to predominate at the stated pH. (a) Alanine at a pH of 10 (b) Proline at a pH of 10 (c) Tyrosine at a pH of 9 (d) Asparagine at physiological pH (e)...
-
Amino acids can be synthesized by reductive amination α-keto of acids (Section 21.8). Biological organisms can also α-keto convert acids into amino acids, but because H2...
-
Do firms follow the same steps for impairment testing of finite- and indefinite- life intangible assets ? Explain.
-
The Deutsche-Telekom board decided against a divestiture of T-Mobile or an initial public offering to pursue a reverse merger. What other alternatives to merging its wholly-owned subsidiary T-Mobile...
-
The data set NBASAL.RAW contains salary information and career statistics for 269 players in the National Basketball Association (NBA). (i) Estimate a model relating points-per-game (points) to years...
-
\(\mathrm{NH}_{3}\) is not always preferred as a refrigerant due to its (a) Flammability (b) Viscosity (c) Toxicity (d) Ozone depletion potential.
-
Sanborn Company has outstanding 40,000 shares of $5 par common stock which had been issued at $30 per share. Sanborn then entered into the following transactions. 1. Purchased 5,000 treasury shares...
-
Mark Down Han Golf Emporium sells golf balls for $27.95 per dozen. The emporium's overhead expenses are 49% of cost and Nahee, the owner, requires a profit of 22% of cost. What is the break-even...
-
1. Are the auditors guilty of malpractice? Explain. 2. What defenses are available to Joker & Wild in this case? Explain what they must prove to successfully assert these defenses. 3. Assume you are...
-
From what amino acid is the following ?-keto acid derived?
-
Identify the following intermediate in the citric acid cycle, and tell whether it has R or Sstereochemistry:
-
Problem refer to the function F in the graph shown. Use the graph to determine whether F'(x) exists at each indicated value of x. x = d F(x) a b c d e f 8 h
-
(a) Explain clearly what a Carbon Tax is. Then explain clearly what an Emissions Trading scheme is. (5 marks) (b) Describe the advantages and disadvantages of a Carbon Tax and Emissions Trading...
-
What are other policy alternatives to extending postpartum Medicaid coverage from 60 days to 12 months after childbirth?
-
Discuss the implications of horizontal gene transfer in prokaryotes on our understanding of evolutionary processes, and how this phenomenon challenges traditional models of phylogenetic tree...
-
College graduation does improve a person's income prospects. But all graduates aren't treate equally: What you majored in counts for a lot. Annual salary surveys by PayScale, Inc., confirm that...
-
In what ways does adaptive radiation contribute to the diversification of phylogenetic lineages in varying ecological niches, and how does this process exemplify the interaction between environmental...
-
Explain the difference between the abiotic features and the biotic features of an organisms environment.
-
Fred Farmer needs to prepare a balance sheet for his bank. He spent the day getting the following information. Fred needs your help to build a balance sheet and evaluate it. The information was...
-
Redo Problem 13.30 using Aspen Plus. Problem 13.30 The liquid in a two-phase, binary mixture of benzene and cyclohexane has a composition of 20 mol % of benzene and 80 mol % of cyclohexane at T =...
-
Name each of the following using IUPAC nomenclature: (a) (CH3)3CCH==CH2 (b) (CH3)2C==CHCH2CH2CH3 (c) (d) CH2-CHCH2CHCHa Cl CH2_CHCH2CHCH3 OH
-
Despite numerous attempts, the alkene 3,4-di-tert-butyl-2,2,5,5- tetramethyl-3-hexene has never been synthesized. Can you explain why? Try making a space-filling model of this compound.
-
Place a double bond in the carbon skeleton shown so as to represent (a) (Z)-1-Methylcyclodecene (b) (E)-1-Methylcyclodecene (c) (Z)-3-Methylcyclodecene (d) (E)-3-Methylcyclodecene (e)...
-
A scuba diver is exploring a shipwreck at a depth of 30 meters in the ocean. The diver's tank contains 5.0 moles of compressed air at a temperature of 25C. What is the pressure of the air in the tank...
-
1. Determine the atmospheric pressure at a location where the barometric reading is 720 mm Hg. 2. A pressure gauge connected to a tank reads 52 psi at a location where the barometric reading is 29.6...
-
Assume that a bond has an annual coupon rate of 6.80 percent, but makes coupon payments on a semi-annual basis, has a par value of $1,000, a yield to maturity of 7.20 percent, and a current price of...

Study smarter with the SolutionInn App


