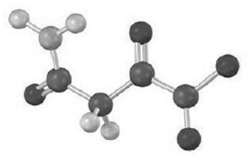
From what amino acid is the following ?-keto acid derived?
Question:
From what amino acid is the following ?-keto acid derived?
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 85% (14 reviews)
As in the previous problem redraw the aketo aci...View the full answer

Answered By

Ali Khawaja
my expertise are as follows: financial accounting : - journal entries - financial statements including balance sheet, profit & loss account, cash flow statement & statement of changes in equity -consolidated statement of financial position. -ratio analysis -depreciation methods -accounting concepts -understanding and application of all international financial reporting standards (ifrs) -international accounting standards (ias) -etc business analysis : -business strategy -strategic choices -business processes -e-business -e-marketing -project management -finance -hrm financial management : -project appraisal -capital budgeting -net present value (npv) -internal rate of return (irr) -net present value(npv) -payback period -strategic position -strategic choices -information technology -project management -finance -human resource management auditing: -internal audit -external audit -substantive procedures -analytic procedures -designing and assessment of internal controls -developing the flow charts & data flow diagrams -audit reports -engagement letter -materiality economics: -micro -macro -game theory -econometric -mathematical application in economics -empirical macroeconomics -international trade -international political economy -monetary theory and policy -public economics ,business law, and all regarding commerce
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The isoelectric point of an amino acid is the pH at which the molecule has no net charge. For glycine, that would be the pH at which virtually all glycine molecules are in the form +H3NCH2CO2-. If we...
-
Amino acid compositions can be determined by heating a protein in 6 M HCl and running the hydrolysate through an ion-exchange column. If you were going to do an amino acid sequencing experiment, why...
-
The amino acid glycine (H2N-CH2-COOH) can participate in the following equilibria in water: (a) Use the values of Ka and Kb to estimate the equilibrium constant for the intramolecular proton transfer...
-
Bookstores International, a bookstore chain, has been quite successful over the past few decades and is now in expansion mode. It typically approaches opening a new bookstore by first determining the...
-
What are the primary disadvantages and advantages of a reverse merger strategy?
-
Use the data in HPRICE1.RAW for this exercise. (i) Estimate the model price = (0 + (1 lotsize + (2 sqrft + (3 bdrms + u and report the results in the usual form, including the standard error of the...
-
Predict the correct one out of the following: (a) The boiling point of a refrigerant should be appreciably lower than the temperature levels at which the refrigerator works (b) The freezing point of...
-
The Ulmer Uranium Company is deciding whether or not it should open a strip mine whose net cost is $4.4 million. Net cash inflows are expected to be $27.7 million, all coming at the end of Year 1....
-
How strongly do you identify with the company's values, mission, and culture, and to what extent does this alignment influence your sense of belonging ?
-
For the analysis and procedure (Step 5 and 6) this needs to be calculated using figure 5. Results for Step 2 (procedure and analysis): Results for Step 3/4: Question 1: Deflection of a beam due to...
-
What -keto acid is formed on transamination of leucine?
-
Identify the amino acid that is a catabolic precursor of each of the following ?-keto acids: (b) (a)
-
Padillas Restoration Company completed the following selected transactions during March 2010: Mar. 1. Established a petty cash fund of $800. 10. The cash sales for the day, according to the cash...
-
Ontario Inc. just paid a $2.00 dividend. Ontario's dividends are expected to grow at 40 percent per year during the next two years, 20 percent over the following year, and then 10 percent per year,...
-
What impact has the lack of credit access or affordable credit had on protected classes? How has the practice of credit discrimination contributed to social injustice in the past? Does this remain a...
-
Company A can borrow at either an 8.5% fixed rate or a floating rate of prime + 1.75% Company B can borrow at either a floating rate of prime + 1.25% or a fixed rate of 8.65% Company A prefers a...
-
What social hierarchies informed Crenshaw's development of the concept of intersectionality in Southern California discussed in class? What legislation did it lead to, and how?
-
1. Importance of change management to organizations. Explanation 2. What is Kotter's Change Model give an overview. Explanation 3. Development of the Kotter's Change Model. Explanation 4. What are...
-
All the organisms that live in a specific area make up a(n) (a) population. (b) community. (c) ecosystem. (d) food chain.
-
The vapor pressure of the liquid NH, is measured at different temperatures. The following vapor pressure data are obtained. Temperature, K P, mmHg 217.1 223.4 234.7 588.1 Calculate the enthalpy of...
-
Redo Problem 13.28 using Aspen Plus. Problem 13.28 A gas mixture containing equimolar quantities of carbon dioxide and hydrogen is to be reformed by passing it over a catalyst. The pressure in the...
-
Write an equation for the reaction of ammonia (:NH3) with hydrogen chloride (HCl). Use curved arrows to track electron movement, and identify the acid, base, conjugate acid, and conjugate base.
-
As noted in Problem 4.7, hydrogen cyanide (HCN) has a pKa of 9.1. Is cyanide ion (CN-) a stronger base or a weaker base than hydroxide ion (HO-)?
-
Write an equation for proton transfer from hydrogen chloride to tert-butyl alcohol. Use curved arrows to track electron movement, and identify the acid, base, conjugate acid, and conjugate base.
-
A cyclist accelerates from rest along straight, horizontal path for a time t accel = 17.5 s at a rate of a = 1.1 m/s squared. part a) Calculate the magnitude of the displacement, in meters, for the...
-
Net Zero Products, a wholesaler of sustainable raw materials, prepares the following aging of receivables analysis. Accounts receivable Percent uncollectible Total $ 153,500 0 $ 91,000 1 to 30 $...
-
The Fibonacci number Fn is recursively defined as F(n) F(n-1)+F(n-2), where F(1) =1 and F(2) = 1. So, F(3) F(2)+F(1)=1+1=2, and so on. Write the MIPS assembly for the fib(n) function, which computes...

Study smarter with the SolutionInn App


