Identify the indicated hydrogen?s in the following molecules as pro-R or pro-S: (a) (b)
Question:
Identify the indicated hydrogen?s in the following molecules as pro-R or pro-S:
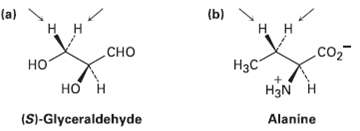
Transcribed Image Text:
(a) н н (b) н н .co2 Сно но Нас но н H3N Alanine (S)-Glyceraldehyde
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 84% (13 reviews)
Strategy For each molecule replace the left hydrogen with 2H Give priorities to ...View the full answer

Answered By

Rustia Melrod
I am a retired teacher with 6 years of experience teaching various science subjects to high school students and undergraduate students. This background enables me to be able to help tutor students who are struggling with the science of business component of their education. Teaching difficult subjects has definitely taught me patience. There is no greater joy for me than to patiently guide a student to the correct answer. When a student has that "aha!" moment, all my efforts are worth it.
The Common Core standards are a useful yardstick for measuring how well students are doing. My students consistently met or exceeded the Common Core standards for science. I believe in working with each student's individual learning styles to help them understand the material. If students were struggling with a concept, I would figure out a different way to teach or apply that concept. I was voted Teacher of the Year six times in my career. I also won an award for Innovative Teaching Style at the 2011 National Teaching Conference.
4.90+
4+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Identify the indicated protons in the following molecules as unrelated, homotopic, enantiotopic, ordiastereotopic: (a) (b) Cysteine
-
Identify the carbon atoms in the following molecules as primary, secondary, tertiary, orquaternary: H H CH CH CHCH2CHCH2CH Gs (c) (a) (b) CH2H2CH CHCCH2CCH CH
-
Label carbon in the following molecules as primary, secondary, tertiary, or quaternary.
-
Peppers Lockdown produces keys for homes and cars. As Peppers is planning for next year's production, he decided to implement a high-low system to forecast future costs. With total production of...
-
Why is it incorrect to say that, when a hot object warms a cold one, the increase in temperature of the cold one is equal to the decrease in temperature of the hot one? When is this statement correct?
-
A simply supported beam ABC is loaded by a vertical load P acting at the end of a bracket BDE (see figure). Draw the shear-force and bending-moment diagrams for beam ABC. IeI 2 4 4
-
Name the six components of an IS.
-
The plaintiff, Herbert Rosenthal Jewelry Corporation, and the defendant, Kalpakian, manufactured jewelry. The plaintiff obtained a copyright registration of a jeweled pin in the shape of a bee....
-
Explain why the circuit in Figure 4 is a 4-to-1 MUX. 85 W W Vo Yo V1 y 12 W2 1 En D D Figure 4 f
-
Using the forecast for the New England Patriots, what is the optimal quantity to order for each player? For blank jerseys? What profit do you expect for Reebok? How much and what type of inventory is...
-
What products are formed from hydration of 4-methylcyclopentene? What can you say about the relative amounts of the products?
-
Identify the indicated faces of carbon atoms in the following molecules as Re orSi: (b) (a) H-2 "CH2 - H Hydroxyacetone Crotyl alcohol
-
A book rests on a table. (a) What is the reaction force to the force the book exerts on the table? (b) To the force gravity exerts on the book?
-
The height in feet of the curved roof of an aircraft hangar can be modeled by y=-0.02x^(2)+1.6x, where x is the distance in feet from one wall at ground leve How tall is the hangar?
-
Purchases Returns and Allowances is what type of account? a. Contra to Equity b. Contra to an Asset O c. Liability O d. Expense Oe. Contra to the cost of Purchases In a manual accounting system,...
-
A yogurt shop offers 4 different flavors of frozen yogurt and 8 different toppings. How many choices are possible for a single serving of frozen yogurt with 1 topping?
-
A ramp for a unloading a moving truck has an angle of elevation of 36o . If the top of the ramp is 5 feet above the ground, approximate the length of the ramp.
-
Judith puts $5000 into an investment account with interest compounded continuously. Which approximate annual rate is needed for the account to grow to $9110 after 30 years?
-
The future of work may look very different from how it does today. The economy has had a significant influence on OB in recent years. The gig economy (also known as freelancing) has arisen as a major...
-
[a] Two foam blocks, each with a charge of 19 micro coulombs (1 C = 10-6 C), are both held in place 19 cm apart in the east-west direction. A foam ball with a charge 49 C is placed 55 cm north of the...
-
How does the approximate root mean square velocity of neon compare to that of krypton at the same temperature? (a) The root mean square velocity of Ne is approximately four times that of Kr. (b) The...
-
When 1,3-diphenyl-2-propanone is treated with Br2 in acid, l,3-dibromo-l,3-diphenyl-2-propanone is obtained in good yield. On further characterization, however, this product proves to have a very...
-
Indicate which hydrogens are replaced by deuterium when each of the following compounds is treated with dilute NaOD in a large excess of CH3OD. (CH) CH-C
-
When compound A in Fig. P22.64 is treated with NaOCH3 in CH3OH, isomerization to compound B occurs. Explain why, when compound C is subjected to the same conditions, no isomerization occurs. NaoCHCH...
-
Larkspur Company sells discounted shoes to the fashion-oriented consumer. The following schedule relates to the company's inventory for the month of March: Cost Sales March 1 Beginning inventory...
-
What is the role of education in promoting social mobility and reducing inequality, and how can educational systems be reformed to better serve diverse populations while preparing individuals for the...
-
A European call option and put option on a non-dividend paying stock both have a strike price of $51 and an expiration date in 6 months. The put sells for $2.50 and the call sells for $2. The...

Study smarter with the SolutionInn App


