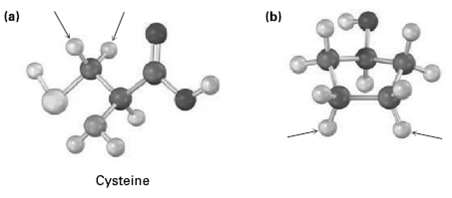
Identify the indicated protons in the following molecules as unrelated, homotopic, enantiotopic, ordiastereotopic: (a) (b) Cysteine
Question:
Identify the indicated protons in the following molecules as unrelated, homotopic, enantiotopic, ordiastereotopic:
Transcribed Image Text:
(a) (b) Cysteine
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (5 reviews)
a Because cysteine has a chirality center the indicated p...View the full answer

Answered By

Charles mwangi
I am a postgraduate in chemistry (Industrial chemistry with management),with writing experience for more than 3 years.I have specialized in content development,questions,term papers and assignments.Majoring in chemistry,information science,management,human resource management,accounting,business law,marketing,psychology,excl expert ,education and engineering.I have tutored in other different platforms where my DNA includes three key aspects i.e,quality papers,timely and free from any academic malpractices.I frequently engage clients in each and every step to ensure quality service delivery.This is to ensure sustainability of the tutoring aspects as well as the credibility of the platform.
4.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Identify the indicated hydrogen?s in the following molecules as pro-R or pro-S: (a) (b) .co2 H3N Alanine (S)-Glyceraldehyde
-
Identify the carbon atoms in the following molecules as primary, secondary, tertiary, orquaternary: H H CH CH CHCH2CHCH2CH Gs (c) (a) (b) CH2H2CH CHCCH2CCH CH
-
Label carbon in the following molecules as primary, secondary, tertiary, or quaternary.
-
The dehydration butanol of alumina is carried out over a silica-alumina catalyst at 680K. CH3CH2CH2CH20H------->cat CH3CH=CHCH3 + H2O The rate law is -r Bu = KPBU/(1+KBuPBul with k= 0.054...
-
Is the following statement correct? "Double marginalization helps firms because it enables them to raise prices."
-
Spinel is a mineral that contains 37.9% Al, 17.1% Mg, and 45.0% O, by mass, and has a density of 3.57 g/cm3. The unit cell is cubic with an edge length of 8.09 . How many atoms of each type are in...
-
Discuss the importance of the verification process and outline its key components.
-
On December 31, 2016, the ledger of Hernandez Company contained the following account balances: All the accounts have normal balances. Journalize the closing entries. Use 4 as the general journal...
-
Address the situation in the media about the 6 police officers that we're fired and charged for murder for beating Tyre Nichols to death. which aspects of criminal law are present and highlighted by...
-
Hallergan Company produces car and truck batteries that it sells primarily to auto manufacturers. Dorothy Hawkins, the company's controller, is preparing the financial statements for the year ended...
-
How many electronically nonequivalent kinds of protons and how many kinds of carbons are present in the following compound? Don?t forget that cyclohexane rings can ring-flip.
-
The following 1H NMR absorptions were obtained on a spectrometer operating at 200MHz and are given in hertz downfield from the TMS standard. Convert the absorptions to units. (a) 436 Hz (b) 956 Hz...
-
Use complex exponentials to prove the identity cos -cos -2 cos
-
Management of WER Limited has decided not to disclose director's' fees in the accounts as they are not material. You cannot convince management to change its decision. However, you do agree that the...
-
February 15, 2024, and you have just arrived at the offices of Weird & Wacky Ltd (WW) which operates a chain of novelty stores across western Canada. WW is owned by the Weasley family and you have...
-
What is the role of system calls in an operating system? Discuss the security implications of system calls and how operating systems mitigate potential vulnerabilities .
-
Discuss the concept of discretionary access control (DAC) versus mandatory access control (MAC) in the context of operating system security. How do these models impact system security and flexibility...
-
11.- : Jordan, age 42, received an eligible rollover distribution of $10,000 from his retirement plan. His employer withheld $2,000 from his distribution. Within 60 days, Jordan decides to contribute...
-
A golf ball is hit from the tee and travels above level ground. Accounting for air resistance, where is the horizontal location of the peak of the flight: at a position that is less than half the...
-
QUESTION 2 The CEO of Farisha Hijab Sdn Bhd insisted on further investigation to be carried out that he also required Mr Muaz to conduct the analysis of variance for the material and labour of the...
-
Calculate the vapor pressure of a solution containing 24.5 g of glycerin (C 3 H 8 O 3 ) in 135 mL of water at 30.0 C. The vapor pressure of pure water at this temperature is 31.8 torr. Assume that...
-
Draw the expected broadband-decoupled 13C NMR spectra of the following compounds. Use Figure 13-41 (page 603) to estimate the chemical shifts. (a) (b) (c) (d) C CH H,C CH C-H C-C
-
(a) Show which carbon atoms correspond with which peaks in the 13C NMR spectrum of butan-2-one (Figure 13-45). (b) Draw the proton NMR spectrum you would expect for butan-2-one. How well do the...
-
Repeat Problem 13-25, sketching the off-resonance-decoupled 13C spectra of the compounds. Problem 13-25 Draw the expected broadband-decoupled 13C NMR spectra of the following compounds. Use Figure...
-
Whirly Corporation's contribution format income statement for the most recent month is shown below: Sales (7,500 units) Variable expenses Contribution margin Fixed expenses Net operating income....
-
After she stopped at the stop sign and looked both ways, Donna pulled onto a roadway and turned right onto Johnson Road.She did not see the Plaintiff's vehicle traveling on Johnson Road. When the...
-
Fuji Co. is growing quickly. Dividends are expected to grow at a rate of 28 percent for the next three years, with the growth rate falling off to a constant 7 percent thereafter. If the required...

Study smarter with the SolutionInn App


