Identify these pairs of compounds as identical, structural isomers, enantiomers, or diastereomers: CH3 CH3 H3 CH Br
Question:
Identify these pairs of compounds as identical, structural isomers, enantiomers, or diastereomers:
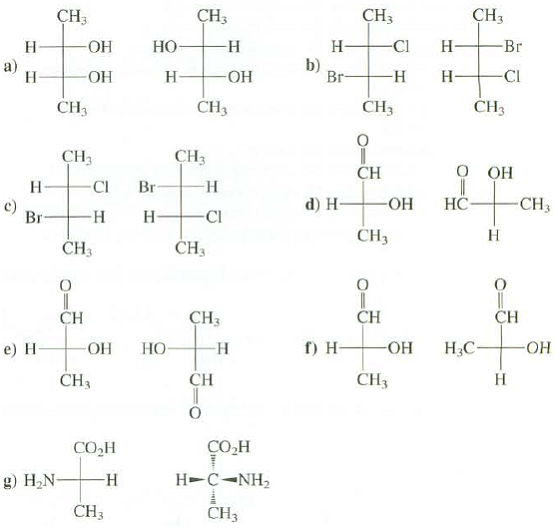
Transcribed Image Text:
CH3 CH3 ÇH3 CHз Br -ОН Но- -н Н- -Cl Н- Н b) Br- a) Н -ОН Н- CI ОН Н- -н CH3 CHз CH3 CH3 CH3 CH3 О он CH Н CI Br -н Но- НС CH3 d) H- c) Br- Н. -Н CI CH3 Н ČH3 CH3 CH CH CH3 CH г) н- НО- Но- Н.С. -ОН -ОН -н e) Н CH CH3 Н CHз СОН СОН Н- с-NH, -н g) Н.N- CH3 CHз
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (12 reviews)
a Diastereomers b D...View the full answer

Answered By

Charles mwangi
I am a postgraduate in chemistry (Industrial chemistry with management),with writing experience for more than 3 years.I have specialized in content development,questions,term papers and assignments.Majoring in chemistry,information science,management,human resource management,accounting,business law,marketing,psychology,excl expert ,education and engineering.I have tutored in other different platforms where my DNA includes three key aspects i.e,quality papers,timely and free from any academic malpractices.I frequently engage clients in each and every step to ensure quality service delivery.This is to ensure sustainability of the tutoring aspects as well as the credibility of the platform.
4.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Identify these pairs of compounds as identical, structural isomers, enantiomers, ordiastereomers: CI b) d) h) i)
-
Identify whether each of following pairs of compounds are enantiomers or diastereomers: a. b. c. d. e. f. OH OH .CI CI
-
Structural isomers are compounds that have the same chemical formula but the atoms are bonded together differently giving different compounds. Consider the two structural isomers having the formula...
-
Unlike other sciences, the nature and value of philosophy is derived from reflection versus testing, measuring, or examining. O True O False 4
-
Friendly Skies Ltd operates the check-in facilities at domestic and international terminals throughout Australia. During the month of May the following costs were incurred as passengers were...
-
On January 1, 2014, Union Power and Light commenced construction of a new generating plant to serve the northeast corridor of the state. The total cost of the project is $ 4,100,000, and it will be...
-
How do you graphically illustrate the System Operations Model?
-
Sunny Day Nursing Home relies heavily on cost data to keep its pricing structures in line with those of its competitors. The facility provides a wide range of services, including assisted living and...
-
what are your opinion regarding Evaluating Database Security and Cyber Attacks paper? here's the link:...
-
Giant pharmaceutical manufacturers such as Merck & Company and Pfizer have traditionally relied heavily on the doctor-to-patient channel to promote their products. In essence, this channel structure...
-
Explain whether or not these compounds would rotate plane-polarized light: ) ) i) OH H C. H3C H.C. HC H HO OH CH3 -OH - CH3 CH3 CH3 CH CH3 b) f) h) HC d) Cl-C-CCI H C "OH H H CH H CHO CH CH3 OH...
-
(a) A solution of 0.2g/mL of a compound in a 1 dm cell rotates plane-polarized light + 13.3 o at the sodium D line. What is the specific rotation of this compound? (b) What is the rotation caused by...
-
a. Find the critical points of the following functions on the domain or on the given interval. b. Use a graphing utility to determine whether each critical point corresponds to a local maximum, local...
-
4. The Cu grains are embedded in a Al2O3 sample forming some grain boundaries. Find the tension on the grain boundary T if the angle theta 0=/3 and interfacial tension Cu/ Al2O3 is 1000mN/m. (10...
-
Starting with double side polished 500 m thick (100) silicon wafers, outline a fabrication process to produce the dovetail joint-type joints shown in the figures. Specifically, outline a fabrication...
-
2. (20 pts) An electric heating element in a residential hot-water heater can be approximated as a D = 10 mm diameter horizontal cylinder with length L = 0.5 m. When powered, the element produces Q =...
-
Load distribution of a wing with finite span. If the load distribution on a finite-span wing is as shown, sketch the trailing vortex system, indicating the magnitude and direction of the trailing...
-
10. Examine the system shown below. Mass 2 sits on mass1 and is free to move on top of mass 1 except there is linear viscous damping with a coefficient of b. Mass 1 is additional mounted to the wall...
-
How can managers plan for changes in the assumptions underlying the budget and manage risk?
-
Southwestern Punch was made by Frutayuda, Inc. and sold in 12-ounce cans to benefit victims of Hurricane Zero. The mean number of ounces placed in a can by an automatic fill pump is 11.7 with a...
-
Solve each inequality and graph its solution. -4n- 7n <0 -7 -6 -5 -5 -4 -3 -4 -3 -2 -1 0 1 2 3
-
What is the relationship between the specific rotations of (2R, 3R)-dichloro- pentane and (2S, 3S)-dichloro pentane? Between (2R, 35)-dichloro pentane and (2R, 3R)-dichloro pentane?
-
What is the stereo chemical configuration of the enantiomer of (2S, 4R)-2, 4-oclanediol?
-
What are the stereo chemical configurations of the two diastereomers of (25, 4R) -2, 4-octanediol?
-
7. An electric charge Q is distributed uniformly throughout a nonconducting sphere of radius Ro with a uniform density, p = Q/{(4/3)R}. Use Gauss's Law to determine (a) (5pts) the electric field,...
-
6.(5pts) A total charge Q is distributed uniformly over a ring of radius R. We found an expression for the value of the electric field on the axis of the ring, E(x), a distance x from the center of...
-
A steel cable that weighs 8 lb/ft is used to pull a 500 lb block of concrete from the ground to the top of a 120 ft tall building. Let x be the distance, in feet, from the block to the TOP of the...

Study smarter with the SolutionInn App


