Identify these pairs of compounds as identical, structural isomers, enantiomers, ordiastereomers: CI b)
Question:
Identify these pairs of compounds as identical, structural isomers, enantiomers, ordiastereomers:
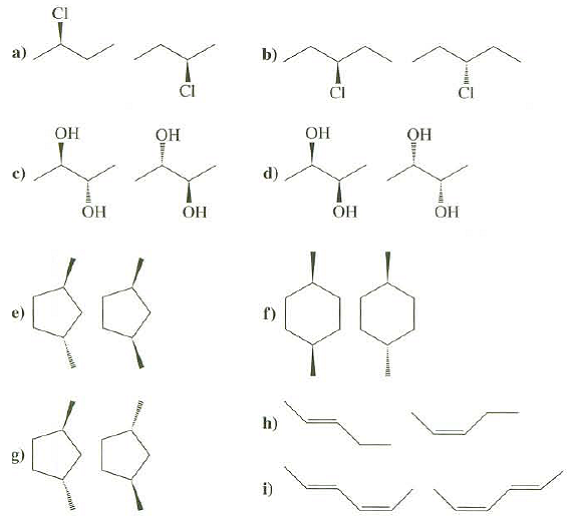
Transcribed Image Text:
CI b) ОН ОН Он Он d) Он ОН ОН ОН h) i)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (15 reviews)
a Identical b Identical ...View the full answer

Answered By

Akshay Agarwal
I am a Post-Graduate with a specialization in Finance. I have been working in the Consulting industry for the past 8 years with a focus on the Corporate and Investment Banking domain. Additionally, I have been involved in supporting student across the globe in their academic assignments and always strive to provide high quality support in a timely manner. My notable achievements in the academic field includes serving more than 10,000 clients across geographies on various courses including Accountancy, Finance, Management among other subjects. I always strive to serve my clients in the best possible way ensuring high quality and well explained solutions, which ensures high grades for the students along-with ensuring complete understanding of the subject matter for them. Further, I also believe in making myself available to the students for any follow-ups and ensures complete support and cooperation throughout the project cycle. My passion in the academic field coupled with my educational qualification and industry experience has proved to be instrumental in my success and has helped me stand out of the rest. Looking forward to have a fruitful experience and a cordial working relationship.
5.00+
179+ Reviews
294+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Identify these pairs of compounds as identical, structural isomers, enantiomers, or diastereomers: CH3 CH3 H3 CH Br - - - - -Cl - b) Br- a) - - CI - - CH3 CH CH3 CH3 CH3 CH3 CH CI Br - - CH3...
-
Identify whether each of following pairs of compounds are enantiomers or diastereomers: a. b. c. d. e. f. OH OH .CI CI
-
Structural isomers are compounds that have the same chemical formula but the atoms are bonded together differently giving different compounds. Consider the two structural isomers having the formula...
-
1. Prepare program using threads in java that can print 10 times the numbers 1,2,3,4,5 in a series. 2. Prepare program using threads and a semaphore in java that can print the numbers 1,2,3,4,5 in a...
-
All Texts and Stationery Ltd (ATS) is a major provider of texts and stationery to secondary schools throughout South Australia. Students submit their order to MS through their school at the beginning...
-
What is a tender offer, and how can it be used as a mechanism to orchestrate a merger?
-
Derive the following expression for determining \(\mathrm{K}_{\mathrm{y}} \mathrm{a}\) from the measurement of \(\mathrm{E}_{\mathrm{MV}}\) in a distillation column if the flow pattern is plug flow....
-
Morgan Companys balance sheet at December 31, 2013, is presented below. During January 2014, the following transactions occurred. (Morgan Company uses the perpetual inventory system.) 1. Morgan paid...
-
2 Required information of 2 No Date January 02, 2024 Cash Answer is not complete. General Journal Common Stock Additional Paid-In Capital 2 February 14, 202 Cash Preferred Stock Additional Paid-In...
-
Recently, Ashland MultiComm Services has been criticized for its inadequate customer service in responding to questions and problems about its telephone, cable television, and Internet services....
-
Explain whether or not these compounds would rotate plane-polarized light: ) ) i) OH H C. H3C H.C. HC H HO OH CH3 -OH - CH3 CH3 CH3 CH CH3 b) f) h) HC d) Cl-C-CCI H C "OH H H CH H CHO CH CH3 OH...
-
(a) A solution of 0.2g/mL of a compound in a 1 dm cell rotates plane-polarized light + 13.3 o at the sodium D line. What is the specific rotation of this compound? (b) What is the rotation caused by...
-
An important task in the audit of the revenue cycle is determining whether a client has appropriately recognized revenue. a. In assessing the risks associated with revenue recognition, the auditor of...
-
Calculate the minimum number of lines needed in a grating that will resolve a doublet of 483.8 and 484.6 nm in the second-order spectrum.
-
Bond A and Bond B are both 2 years from maturity, have a 5% YTM, and pays semiannually. Bond A has a 5% coupon rate, Bond B has a 9% coupon rate. Calculate the duration of Bond A.
-
Analyze the role of advanced soil mechanics and foundation engineering techniques in land development projects, including the use of numerical modeling methods like finite element analysis to assess...
-
16. Calculate the present value of the annuity. (Round your answer to the nearest cent.)$11,000 annually at 5% for 10 years.
-
A piston and shaft moving through an oil bath is used to reduce the linear speed of an impact testing machine bar. The velocity of the piston is v= 2 m/s at A, and it moves with a deceleration of a =...
-
Derive the frequency equation of the system shown in Fig. 6.29. k k k3 KA 000 m 000 m2 000 m3 000 FIGURE 6.29 Spring-mass system.
-
Why do CPA firms sometimes use a combination of positive and negative confirmations on the same audit?
-
Solve each inequality and graph its solution. 16 -v + 7 81 28
-
Which of the following objects arc chiral? (a) A basketball (b) A fork (c) A wine glass (d) A golf club (e) A monkey wrench (f) A snowflake
-
Erythronolide B is the biological precursor of erythromycin, a broad-spectrum antibiotic. How many chirality centers does Erythronolide Bhave? , .C H . CH3 . Erythronolide B . , . CH
-
Draw examples of the following: (a) A meso compound with the formula C8H18 (b) A meso compound with the formula C9H20 (c) A compound with two chirality centers, one R and the other S
-
For the given cash flows, suppose the firm uses the NPV decision rule. Year Cash Flow 160,000 0 -$ 56,000 123 83,000 67,000 a. At a required return of 9 percent, what is the NPV of the project? (Do...
-
New-Project Analysis The Campbell Company is considering adding a robotic paint sprayer to its production line. The sprayer's base price is $800,000, and it would cost another $20,000 to install it....
-
Question 5 0 / 5 pts What is the expected market value of the following bonds in the ML Brokerage Account if interest rates decrease by 1.41%? Assume interest is semiannually. $50,000 US Treasury...

Study smarter with the SolutionInn App


