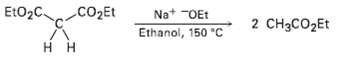
In contrast to the rapid reaction shown in Problem 23.40, ethyl acetoacetate requires a temperature over 150
Question:
In contrast to the rapid reaction shown in Problem 23.40, ethyl acetoacetate requires a temperature over 150 ?C to undergo the same kind of cleavage reaction. How can you explain the difference in reactivity?
Transcribed Image Text:
EtO2C CO2Et Na* "OEt Ethanol, 150 C 2 CH3CO2Et H H
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 63% (11 reviews)
Two different reactions are possible when ethyl acetoacetate reacts with etho...View the full answer

Answered By

Shubhradeep Maity
I am an experienced and talented freelance writer passionate about creating high-quality content. I have over five years of experience working in the field and have collaborated with several renowned companies and clients in the SaaS industry.
At Herman LLC, an online collective of writers, I generated 1,000+ views on my content and created journal content for 100+ clients on finance topics. My efforts led to a 60% increase in customer engagement for finance clients through revamping website pages and email interaction.
Previously, at Gerhold, a data management platform using blockchain, I wrote and published over 50 articles on topics such as Business Finance, Scalability, and Financial Security. I managed four writing projects concurrently and increased the average salary per page from $4 to $7 in three months.
In my previous role at Bernier, I created content for 40+ clients within the finance industry, increasing sales by up to 40%.
I am an accomplished writer with a track record of delivering high-quality content on time and within budget. I am dedicated to helping my clients achieve their goals and providing exceptional results.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
In contrast to the reaction with dilute alkali (Section 18.6), when concentrated solutions of NaOH are used, acetoacetic esters undergo cleavage as shown below.
-
In contrast to the United States, Japan has realized continuous current account surpluses. What could be the main causes for these surpluses? Is it desirable to have continuous current account...
-
In contrast to the capital asset pricing model, arbitrage pricing theory: a. Requires that markets be in equilibrium. b. Uses risk premiums based on micro variables. c. Specifies the number and...
-
A Monica who is aged 54, is employed as a Senior Manager with Abacus, a large accounting firm based in Sydney. During the 2020 tax year Abacus contributed $20,000 into QRest Super, which Monica...
-
Two incompatible high-resolution audio formats, Super Audio CD (SACD) and DVD Audio (DVDA), were introduced in 2000. Both offer surround-sound music at a quality that approaches the original studio...
-
As shown in the accompanying photograph, an electric stove burner on its highest setting exhibits an orange glow. (a) When the burner setting is changed to low, the burner continues to produce heat...
-
What does an organization need to put in place to identify suitable strategic technologies?
-
Gordeeva Corporation began selling goods on the installment basis on January 1, 2010. During 2010, Gordeeva had installment sales of $150,000; cash collections of $54,000; cost of installment sales...
-
How can the presence of subcultures within a larger organizational culture affect both alignment with corporate values and overall organizational performance?
-
Phenol OH groups have an approximate pka of 10, while NaHCO3 (the conjugate acid of Na2CO3) has a pka of 10.3. This means the phenol and it's conjugate base (phenoxide) should exist in equilibrium...
-
Ethyl dimethylacetoacetate reacts instantly at room temperature when treated with ethoxide ion to yield two products, ethyl acetate and ethyl 2-mcthylpropanoatc. Propose a mechanism for this...
-
How might the following compounds be prepared using Michael reactions? Show (he nucleophilic donor and the electrophilic acceptor in eachcase. ", , (b) . (a) CHCCH-CH2CH2CH3 CH2H2CCgHs o2Et NO2 (d)...
-
Find the domain of the function. Do not use a graphing calculator. x7 - zx = (x)
-
Jack throws a squirrel off of a building with an initial speed of 4 m/s down. If the squirrel takes 0.75 s to reach the ground, a) How tall is the building (how far does the squirrel fall)? b) How...
-
The following are selected accounts and balances for Jonah Company and Hill, Incorporated, as of December 31, 2024. Several of Jonah's accounts have been omitted. Credit balances are Indicated by...
-
Primare Corporation provided the following data for last month s manufacturing operations. Purchases of raw materials $ 3 0 , 0 0 0 Indirect materials used in production $ 4 , 9 6 0 Direct labor $ 5...
-
A Company borrowed money from a local bank. The note the company signed requires five annual installment payments of $16,500 not due for three years. The interest rate on the note is 7%. What amount...
-
Tanya is 49 years of age and her 2023 income is made up of employment income of $88,500. She contributed $9,300 to her Registered Retirement Savings Plan in 2023. (Assuming she has the RRSP...
-
A small puck on an air table revolves in a circle with rotational speed \(\omega\), held at radius \(r\) by a weighted string that passes through a hole in the table. You slowly pull down on the...
-
The Thomas Corporation was organized on Jan. 1, 2020. On Dec. 31, 2021, the corporation lost most of its inventory in a warehouse fire before the year-end count of inventory was to take place. just...
-
Write the Lewis structure for each molecule or ion. a. N 2 H 2 b. N 2 H 4 c. C 2 H 2 d. C 2 H 4
-
Determine which compounds show cis-trans isomerism. Draw and label the isomers, using both the cis-trans and E-Z nomenclatures where applicable. (a) pent-1-ene (b) pent-2-ene (c) hex-3-ene (d)...
-
For each alkene, indicate the direction of the dipole moment. For each pair, determine which compound has the larger dipole moment. (a) Cis-1,2-difluoroethene or trans-1,2-difluoroethene (b)...
-
Predict the products of the following reactions. When more than one product is expected, predict which will be the major product. (a) (b) (c) (d) OH H2SO4 heat H3PO4 heat BrNaocH, CH CH H,SO4 heat OH
-
Calculate the energy stored for the series-parallel combination shown in figure if C1 = 2uF, C = 1.6F, C3 = 4.3F, C = 6F, and C5 = 8F and charged to a potential difference of 3200- V. (11pnts) C3 HH...
-
Comprehensively explains the financial cost of a total reward plan that incudes improvements in the health and wellness plan, 401(K) plan and a sick bank to both direct and indirect compensation ?
-
A company that uses job order costing purchases $86,000 in raw materials for cash. It then uses $12,000 of raw materials as indirect materials and uses $67,700 of raw materials as direct materials....

Study smarter with the SolutionInn App


