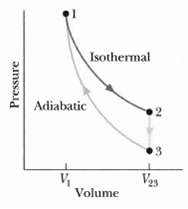
In Figure where V23 = 3.00V1, n moles of a diatomic ideal gas are taken through the
Question:
In Figure where V23 = 3.00V1, n moles of a diatomic ideal gas are taken through the cycle with the molecules rotating but not oscillating. What are?
(a) p2/p1
(b) p3/p1 and
(c) T3/T1 For path 1 ?? 2, what are?
(d) W/nRT1,
(e) Q/nRT1,
(f) ΔEint/nRT1, and
(g) ΔS/nR? For path 2 ?? 3, what are?
(h) W/nRT1
(i) Q/nRT1,
(j) ΔEint/nRT1,
(k) ΔS/nR? For path 3 ?? L, what are?
(l) W/nRT1,
(m) Q/nRT1,
(n) ΔEint/nRT1,
(o) ΔS/nR?
Transcribed Image Text:
Isothermal Adiabatic Vas Volume Pressure
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 64% (17 reviews)
The connection between molar heat capacity and the degrees of freedom of a diatomic gas is given by setting f 5 in Eq 1951 Thus Cy5R2 C7R2 and y 75 In ...View the full answer

Answered By

Vineet Kumar Yadav
I am a biotech engineer and cleared jee exam 2 times and also i am a math tutor. topper comunity , chegg India, vedantu doubt expert( solving doubt for iit jee student on the online doubt solving app in live chat with student)
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Fundamentals of Physics
ISBN: 978-0471758013
8th Extended edition
Authors: Jearl Walker, Halliday Resnick
Question Posted:
Students also viewed these Thermodynamics questions
-
A beat engine takes 0.350 mol of a diatomic ideal gas around the cycle shown in the pV-diagram of fig. 20.24. Process I 2 is at constant volume, process 23 is adiabatic, and process 3 1 is at a...
-
Two moles of a diatomic ideal gas are compressed isothermally from 18 L to 8 L. In the process, 170 calories escape from the system. Determine the amount of work done by the gas, the change in...
-
A 1.00-mol sample of a diatomic ideal gas has pressure P and volume V. When the gas is heated, its pressure triples and its volume doubles. This heating process includes two steps, the first at...
-
In the light of what you know about IHRM, how easy do you think it is to manage a global employer brand? What issues would an IHRM professional expect to have to manage? To answer this, analyse the...
-
Using the Stolper-Samuelson theorem, indicate the effect on the distribution of income between labor and capital in Nation 1 (assumed to be a small nation) when it imposes an import tariff on...
-
Andrew Manufacturing held an average inventory of $1.1 million (raw materials, work-in-process, finished goods) last year. Its sales were $8.0 million, and its cost of goods sold was $5.8 million....
-
Why does average employee contribution offer a better estimate of the contribution of individuals to an organization than total compensation (wages, incentives, and benefits)?
-
One item is omitted in each of the following summaries of balance sheet and income statement data for four different corporations, AL, CO, KS, and MT. Determine the missing amounts, identifying them...
-
Image transcription text Question 6 Not yet answered Marked out of 1.00 '1" Flag question During a mountain?biking tripr Bill and Barry are discussing the merits of having the springs and shocks used...
-
Sharon Gillespie, a new buyer at Visions, Inc., was reviewing quotations for a tooling con. tract submitted by four suppliers. She was evaluating the quotes based on price, target quality levels. and...
-
At very low temperatures, the molar specific heat Cv of many solids is approximately Cv = AT3, where A depends on the particular substance. For aluminuffi, A = 3.15 X 10-5 J/mol K4. Find the entropy...
-
A 2.0 mol sample of an ideal monatomic gas undergoe the reversible process shown in Figure, the scale of the vertical axis is set by Ts = 400.0K and the scale of the horizontal axis is set by Ss =...
-
Ozark University admission office is processing new applications for the upcoming academic year. The applications fall into three categories: in-state, out-of-state, and international. The malefemale...
-
a.[2pts] Create a 1-variable Data table where the input is the sales growth rate, and the output is the NPV. The sales growth rate should increase from 7.5% to 12% with a constant increment of 0.5%....
-
- You decided to open a production facility. The following figure shows the process flow diagram. You hired 5 employees, and each employee performs one task. A-How many units can you produce in 10...
-
Check whether the sections shown in Figure 3 are underreinforced or not. If underreinforced, calculate the ultimate moment capacities. Materials are C20 and S420. Use material factors Yms, Ymc. Take...
-
In March 31, 2021, the end of the fiscal year, the preliminary Trial Balance of MSL Manufacturing Company showed the following information: Accounts Payable Accounts Receivable 375,000 500,000...
-
A distributor of industrial equipment purchases specialized compressors for use in air conditioners. The regular price is $45, however, the manufacturer of this compressor offers quantity discounts...
-
Trevs Gardening Services purchased a trailer on 1 July 2019 for $26 200. It was estimated to have a useful life of 5 years and a residual value at the end of that time of $2800. Required (a) What is...
-
Estimate a range for the optimal objective value for the following LPs: (a) Minimize z = 5x1 + 2x2 Subject to X1 - x2 3 2x1 + 3x2 5 X1, x2 0 (b) Maximize z = x1 + 5x2 + 3x3 Subject to X1 + 2x2 +...
-
A 0.0250-kg bullet is accelerated from rest to a speed of 550 m/s in a 3.00-kg rifle. The pain of the rifles kick is much worse if you hold the gun loosely a few centimeters from your shoulder rather...
-
In Example 16.19 (Section 16.8), suppose the police car is moving away from the warehouse at 20 m/s. What frequency does the driver of the police car hear reflected from the warehouse?
-
Two train whistles, A and B, each have a frequency of 392 Hz. A is stationary and B is moving toward the right (away from A) at a speed of35.0 m/s. A listener is between the two whistles and is...
-
A railroad train is traveling at 25.0 m/s in still air. The frequency of the note emitted by the locomotive whistle is 400 Hz. What is the wavelength of the sound waves? (a) In front of the...
-
A retirement home in Florida costs $ 2 0 0 , 0 0 0 today. Housing prices in Florida are increasing at a rate of 4 % per year. Joe wants to buy the home in 8 years when he retires. Joe has $ 2 5 , 0 0...
-
1. (55 points) Answer the following questions about the steady state system shown below. (Don't worry about starting from 1st Law - use common engineering assumptions.) a) Is it appropriate to use...
-
Describe at least three ways that Guidance Software responds to and helps to manage changes in our technology - driven world? Describe what forecasting involves and list three limitations to...

Study smarter with the SolutionInn App


