Isoleucine and threonine are the only two amino acids with two chirality centers. Assign R or S
Question:
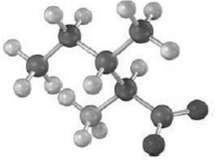
Isoleucine and threonine are the only two amino acids with two chirality centers. Assign R or S configuration to the methyl-bearing carbon atom ofisoleucine.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 85% (7 reviews)
HH C...View the full answer

Answered By

Sumit kumar
Education details:
QUATERNARY Pursuing M.Tech.(2017-2019) in Electronics and Communication Engg. (VLSI DESIGN) from
GNIOT Greater Noida
TERTIARY B.Tech. (2012-2016) in Electronics and Communication Engg. from GLBITM Greater Noida
SECONDARY Senior Secondary School Examination (Class XII) in 2012 from R.S.S.Inter College, Noida
ELEMENTARY Secondary School Examination (Class X) in 2010 from New R.J.C. Public School ,Noida
CERTIFICATION
Summer Training in ‘WIRELESS EMBEDDED SYSTEM’ from ‘XIONEE’ for the six weeks.
EMBEDDED SYSTEM Certificate issued by CETPA INFOTECH for one day workshop.
Certificate of Faculty development program on OPTICAL COMMUNICATION and NETWORKS for one week.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Assign R or S configuration to the chirality center in the following molecular model of the amino acid methionine (blue = N, yellow =S):
-
Assign R or S configuration to each chirality center in the followingmolecules: (a) (b) ,CH (c) , . CH CHCH
-
Assign R or S configuration to each chirality center in the following biologicalmolecules: (a) (b) .C 'N' N-N - CH2CH2CH2CH2CO2 Prostaglandin E, Biotin
-
Consider a portfolio of the following derivatives where the counterparty is an OECD bank. Derivative 8-year interest rate swap 6-month option on an equity 1-year swap on precious metals 9-month...
-
Production on a popular line of decorative lighting appliances produced at your Chinese manufacturing plant inexplicably came to a halt last month. As the product manager in the United States, you...
-
How could you use IR spectroscopy to distinguish between the isomers 1-hexyne and 1,3-hexadiene?
-
Design a 1-4 shell-and-tube heat exchanger to cool \(60,000 \mathrm{lb} / \mathrm{hr}\) of \(42^{\circ} \mathrm{API}\) kerosene from 400 to \(220^{\circ} \mathrm{F}\) by heating a \(35^{\circ}...
-
Southern Copper Corporation (PCU) acquired mining equipment for $100,000 cash on January 1, 2009. The equipment had an expected useful life of four years and zero salvage value . PCU calculates...
-
If following code was executed, what is the final value of variable y? y=2; for k=0: 3 y = y +2*k; end
-
Today is May 15, 2000, and the current, semi-annually compounded yield curve is in Table 3.6. Compute the duration for the following securities: (a) 3-year zero coupon bond (b) 3 1/4-year coupon bond...
-
Give the sequence of the following tetra peptide (yellow =S):
-
Is the following structure a D amino acid or an L amino acid? Identifyit.
-
When 26.5 mL of 0.100 M Ca(NO 3 ) 2 is combined with 49.8 mL of 0.100 M NaF, (a) What is the theoretical yield of CaF 2 (s) in grams? (b) What is the molar concentration of Ca 2+ (excess reactant) in...
-
Section 7.3 Stockholders' Approval. (a) Target shall promptly call a meeting of its stockholders for the purpose of voting upon this Agreement and the transactions contemplated hereby and, subject to...
-
For each of the following, illustrate with suitable solution function graphs and/or phase lines the way in which the solutions change as the value of r changes. Identify the precise value(s) of r for...
-
The WBS can be formatted in an indented outline, an organizational chart, and a free format. Discuss the strengths and weaknesses of each of these three formats. In addition, explain which format you...
-
1) Suppose a town's population is composed of 3 types of voters with the following distribution: 20% are in Group 1, 35% are in Group 2, and 45% are in Group 3. The town wants to build a park. They...
-
A certain life insurance company is seeking to improve csutomer experience by automating the life insurance application process. As a business analyst, how do you develop a business analysis plan...
-
In Problem 124, how much will be in the fund immediately after the thirtieth deposit if the fund pays 6 percent compounded annually and each successive deposit is 6 percent greater than the preceding...
-
Decades after the event, Johnson & Johnson (J&J), the 130-year-old American multinational, is still praised for swiftly The company indicated that its response was based on the expectations set forth...
-
Which is a stronger base, S 2- or Se 2- ? Explain.
-
Devise a synthesis of 4-methylphenyl propyl ketone from toluene and butanoic acid as starting materials.
-
Write the structural formula for a. Butanoic anhydride b. Benzoic anhydride
-
Predict and name the product of the following reaction: COOH heat COOH
-
1. A car accelerates from rest to 16.21 meters per second in 7.66 seconds. What is the car's rate of acceleration in meters per second squared? Round your answer to two decimal places. 2. If the...
-
The debits to Work in Process-Roasting Department for Morning Brew Coffee Company for August, together with information concerning production, are as follows: Work in process, August 1, 800 pounds,...
-
Solve these equations Sy = b with 81, 82, 83 in the columns of S: 0 0 10 Y2 100 0 1998-8-1988-8 and S is a sum matrix. The sum of the first 5 odd numbers is ][

Study smarter with the SolutionInn App


